Chapter: Obstetrics and Gynecology: Common Medical Problems in Pregnancy
Cardiac Disease during pregnancy
CARDIAC DISEASE
With earlier diagnoses and more
effective treatments, more women with congenital and acquired heart disease
reach adulthood and may become pregnant. Patients with rheumatic heart disease
(caused by untreated or delayed treatment of group A–hemolytic streptococcal
[GAS] ton-sillopharyngitis) and acquired infectious valvular heart dis-ease
(often associated with drug use) comprise only 50% of pregnant cardiac
patients. The remainder consists of other cardiac conditions that traditionally
have been less commonly seen in pregnancy. Because
pregnancy is itselfassociated with an increase in cardiac output of 40%, the
risks to mother and fetus are often profound for women with pre-existing
cardiac disease. Ideally, cardiac patients should havepreconceptional care
directed at maximizing cardiac func-tion. They should also be counseled about
the risks their particular heart disease poses in pregnancy.
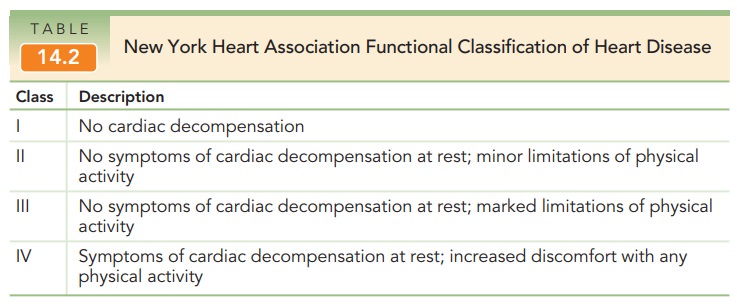
Classification of Heart Diseases in Pregnancy
The classification of heart
disease by the New York Heart Association is useful to evaluate all types of
cardiac patients with respect to pregnancy (Table 14.2). It is a functional
classification and is independent of the type of heart dis-ease. Patients with
septal defects, patent ductus arteriosus, and mild mitral and aortic valvular
disorders often are in classes I or II and tend to do well throughout
pregnancy. Primary pulmonary
hypertension, uncorrected tetralogy of Fallot, Eisenmenger syndrome, Marfan
syndrome with significant aortic root dilation, and certain other conditions
are associated with a much worse prognosis (frequently death) through the
course of pregnancy. For this reason, patients with such disorders are strongly
advised not to become pregnant.
Management
General management of the pregnant cardiac patient con-sists of avoiding conditions that add additional stress to the workload of the heart beyond that already imposed by preg-nancy, including prevention and/or correction of anemia, prompt recognition and treatment of any infections, a de-crease in physical activity and strenuous work, and proper weight gain. Adequate rest is essential. For patients with class I or II heart disease, increased rest at home is advised; and in cases of higher class levels, hospitalization and treatment of cardiac failure may be required. Coordinated management between obstetrician, cardiologist, and anes-thesiologist is especially important for patients with sig-nificant cardiac dysfunction.
The fetuses of patients with
functionally significant cardiac disease are at increased risk for low birth
weight and prematurity. A patient with
congenital heart disease is1% to 5% more likely to have a fetus with congenital
heart disease than is someone without this condition; antepartum fetal cardiac
assessment using ultrasound is recommended.
The antepartum management of
pregnant cardiac pa-tients includes serial evaluation of maternal cardiac
status as well as fetal well-being and growth. Anticoagulation, antibiotic
prophylaxis for subacute bacterial endocarditis, invasive cardiac monitoring,
and even surgical correction of certain cardiac lesions during pregnancy can
all be ac-complished if necessary. The intrapartum and postpartum management of
pregnant cardiac patients includes consid-eration of the increased stress of
delivery and postpartum physiologic adjustment. Labor in the lateral position
to fa-cilitate cardiac function is often desirable. Every attempt is made to
facilitate vaginal delivery because of the in-creased cardiac stress of
cesarean section. Because cardiac output increases by 40% to 50% during the
second stage of labor, shortening this stage by the use of forceps or vac-uum
extractor is often advisable. Epidural anesthesia to re-duce the stress of
labor is also recommended. Even with patients who are stable at the time of
delivery, cardiac out-put increases in the postpartum period because of the
ad-ditional 500 mL added to the maternal blood volume as the uterus contracts.
Indeed, most obstetric patients who die with cardiac disease do so following
delivery.
Rheumatic
heart disease remains a common cardiacdisease in pregnancy. As the severity of the associated valvu-lar
lesion increases, the risk for thromboembolic disease, subacute bacterial
endocarditis, cardiac failure, and pulmonary edema increases. A high rate of
fetal loss also occurs in women with rheumatic heart disease. Approximately
90% of these pa-tients have mitral stenosis, whose associated mechanical
obstruction worsens as cardiac output increases during pregnancy. Women with
mitral stenosis associated with atrial fibrillation have an especially high
risk of develop-ing congestive heart failure.
Maternal
cardiac arrhythmias are occasionally en-countered
during pregnancy. Paroxysmal atrial
tachycardia is the most commonly encountered maternal arrhythmia and is usually
associated with overly strenuous exercise. Underlyingcardiac disease such
as mitral stenosis should be suspected when atrial fibrillation and flutter are
encountered.
Peripartum
cardiomyopathy is an unusual but espe-cially severe cardiac
condition identified in the last month of pregnancy or the first 6 months
following delivery. It is difficult to distinguish from other cardiomyopathies
(e.g., myocarditis) except for its association with pregnancy. In many cases,
no apparent cause can be determined. Treatment
is generally unchanged from cardiac failure un-associated with pregnancy,
except that the use of angiotensin-converting enzyme inhibitors is avoided if
the patient is pregnant. Management includes bed rest, digoxin, diuretics,
and, in some cases, anticoagulation. The mortality rate is high and is related
to cardiac size 6 to 12 months later. If cardiac size returns to normal,
prognosis is improved, although it remains guarded. Sterilization counseling is
warranted for patients with cardiomyopathy.
Related Topics