Chapter: Biochemical Pharmacology : Some principles of cancer pharmacotherapy
Antibiotics
Antibiotics
Another class of DNA-damaging
drugs are found in nature as antibiotics. Antibiotics are widely used by
compet-ing soil microorganisms, both prokaryotic and eukaryotic. While many of
them are selectively toxic for prokaryotic cells and therefore of use in
antibacterial therapy, an even larger number is toxic for both prokaryotes and
eukaryotes, often with higher toxicity for the latter. Several of these
an-tibiotics have been useful in biochemical research because they can be used
to dissect various stages of gene expres-sion and protein maturation.
The
antibiotics used in cancer chemotherapy are most-ly DNA-intercalating agents.
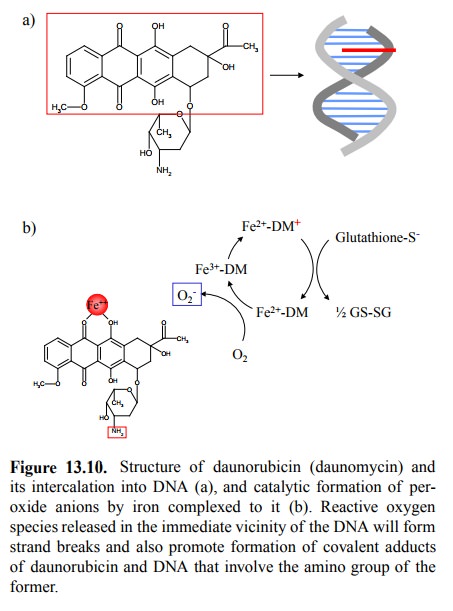
An example is provided by daunorubicin (Figure 13.10). This drug molecule has a
large, flat polycyclic system, which will intercalate between the stacked base
pairs of the DNA. However, there is more to it: The ring is also able to
chelate iron, and the complex catalyses the formation of superoxide anions at
the expense of glutathione (Figure 13.10b). The highly reactive super-oxide can
wreak all kinds of havoc on the DNA in its im-mediate vicinity, including
strand breakages10. Another ef-fect is the formation of covalent
adducts between the amino group at the sugar moiety and guanine residues in the
DNA. In the chapter on drug metabolism, we already saw some-thing similar: The
intercalating agent benzpyrene also re-quired covalent reactivity for
significant DNA damage to occur, which in that case was brought about by
metabolic activation to the epoxide.
Related Topics