Chapter: Clinical Anesthesiology: Anesthetic Management: Obstetric Anesthesia
Anesthesia for Cesarean Section: General Anesthesia
GENERAL ANESTHESIA
Pulmonary aspiration of gastric contents and
failed endotracheal intubation are the major causes of maternal morbidity and mortality
associated with general anesthesia. All patients should receive pro-phylaxis
against aspiration pneumonia with 30 mL of 0.3 M sodium citrate 30–45 min prior
to induc-tion. Patients with additional risk factors pre-disposing them to
aspiration should also receive intravenous ranitidine, 50 mg, or
metoclopramide, 10 mg, or both, 1–2 h prior to induction; such fac-tors include
morbid obesity, symptoms of gastro-esophageal reflux, a potentially difficult
airway, or emergent surgical delivery without an elective fast-ing period.
Premedication with oral omeprazole, 40 mg, at night and in the morning also
appears to be highly effective in high-risk patients undergoing elective
cesarean section. Although anticholinergics theoretically may reduce lower esophageal
sphinc-ter tone, premedication with glycopyrrolate (0.1 mg) helps reduce airway
secretions and should be con-sidered in patients with a potentially difficult
airway.
Anticipation of a difficult endotracheal
intuba-tion may help reduce the incidence of failed intuba-tions. Examination
of the neck, mandible, dentition, and oropharynx often helps predict which
patients may have problems. Useful predictors of a diffi-cult intubation
include Mallampati classification, short neck, receding mandible, prominent
maxil-lary incisors, and history of difficult intubation . The higher incidence
of failed intuba-tions in pregnant patients compared with nonpreg-nant surgical
patients may be due to airway edema, a full dentition, or large breasts that
can obstruct the handle of the laryngoscope in patients with short necks.
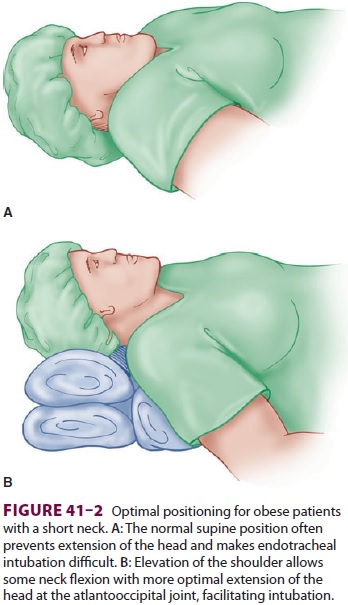
Proper positioning of the head and neck may facilitate endotracheal intubation
in obese patients: elevation of the shoulders, flexion of the cervical spine,
and extension of the atlantooccipital joint (Figure 41–2). A variety of laryngoscope blades, a short laryngoscope handle, at
least one extra stiletted endotracheal tube (6 mm), Magill forceps (for nasal
intubation), a laryngeal mask airway (LMA), an intu-bating LMA (Fastrach), a
fiberoptic bronchoscope,
a video-assisted laryngoscope (GlideScope or
Stortz CMAC), the capability for transtracheal jet ventila-tion, and possibly
an esophageal–tracheal Combitube should be readily available .
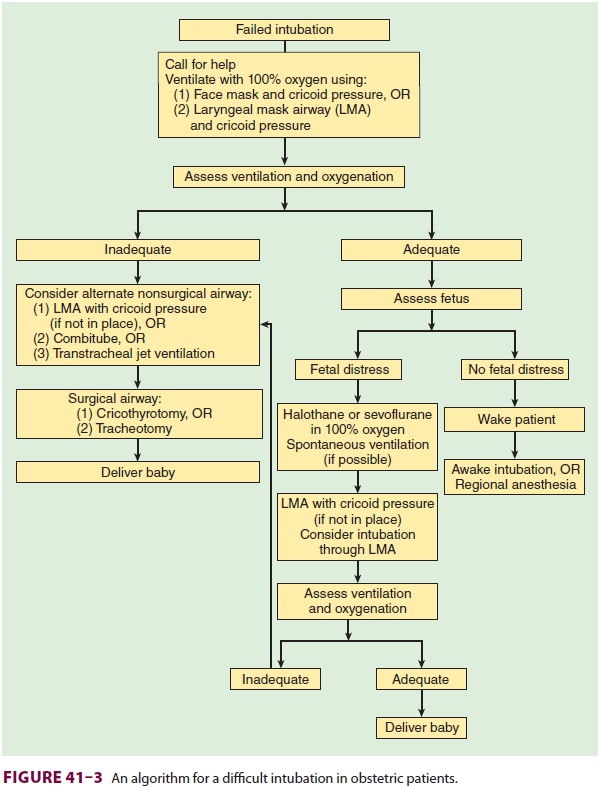
When potential difficulty in securing the
air-way is suspected, alternatives to the standard rapid-sequence induction
with conventional laryngoscopy, such as regional anesthesia or awake fiberoptic
tech-niques, should be considered. We have found that video-assisted laryngoscopy
has greatly reduced the incidence of difficult or failed tracheal intubation at
our institutions. Moreover, a clear plan should be formulated for a failed
endotracheal intubation following induction of anesthesia (Figure
41–3). In the absence of fetal distress, the
patient should be awakened, and an awake intubation, with regional or local
(infiltration) anesthesia, may be tried. In the presence of fetal distress, if
spontaneous or positive-pressure ventilation (by mask or LMA) with cricoid
pressure is possible, delivery of the fetus may be attempted. In such
instances, a potent volatile agent with oxygen is employed for anesthesia, but
once the fetus is delivered, nitrous oxide may be added to reduce the
concentration of the volatile agent; sevo-flurane may be the best volatile
agent because it may be least likely to depress ventilation. The inability to
ventilate the patient at any time may require imme-diate cricothyrotomy or
tracheostomy.
Suggested Technique for Cesarean Section
·
The patient is placed
supine with a wedge under the right hip for left uterine displacement.
·
Denitrogenation is
accomplished with 100% oxygen for 3–5 min while monitors are applied.
·
The patient is prepared
and draped for surgery.
·
When the surgeons are
ready, a rapid-sequence induction with cricoid pressure is performed using
propofol, 2 mg/kg, or ketamine,
·
1–2 mg/kg, and succinylcholine, 1.5
mg/ kg. Ketamine is used instead of propofol in hypovolemic patients. Other
agents, including methohexital and etomidate, offer little benefit in obstetric
patients.
·
With few exceptions,
surgery is begun only after proper placement of the endotracheal tube is
confirmed. Excessive hyperventilation (Paco2 25 mm Hg) should be
avoided because it can reduce uterine blood flow and has been associated with
fetal acidosis.
· Fifty percent nitrous oxide in oxygen with up to 0.75 MAC of a low concentration of volatile agent (eg, 1% sevoflurane, 0.75% isoflurane, or 3% desflurane) is used for maintenance of anesthesia. The low dose of volatile agent helps ensure amnesia but is generally not enough to cause excessive uterine relaxation or prevent uterine contraction following oxytocin. A muscle relaxant of intermediate duration (atracurium, cisatracurium, or rocuronium) is used for relaxation, but may exhibit prolonged neuromuscular blockade in patients who are receiving magnesium sulfate.
·
After the neonate and
placenta are delivered, 20–80 units of oxytocin are added to the first liter of
intravenous fluid, and another 20 units to the next. Additional intravenous
agents, such as propofol, opioid, or benzodiazepine, can be given to ensure
amnesia.If the uterus does not contract readily, an opioid should be given, and
the halogenated agent should be discontinued. Methylergonovine (Methergine),
0.2 mg intramuscularly or in 100-mL normal saline as slow intravenous infusion,
may also begiven but can increase arterial blood pressure.
15-Methylprostaglandin F2α (Hemabate), 0.25 mg intramuscularly, may
also be used.
·
An attempt to aspirate gastric
contents may be made via an oral gastric tube to decrease the likelihood of
pulmonary aspiration on emergence.
·
At the end of surgery,
muscle relaxants are completely reversed, the gastric tube (if placed) is
removed, and the patient is extubated while awake to reduce the risk of
aspiration.
Related Topics