Chapter: Plant Biology : Metabolism
Amino acid, lipid, polysaccharide and secondary product metabolism
AMINO ACID, LIPID, POLYSACCHARIDE AND SECONDARY PRODUCT METABOLISM
Key Notes
Role of the citric acid cycle
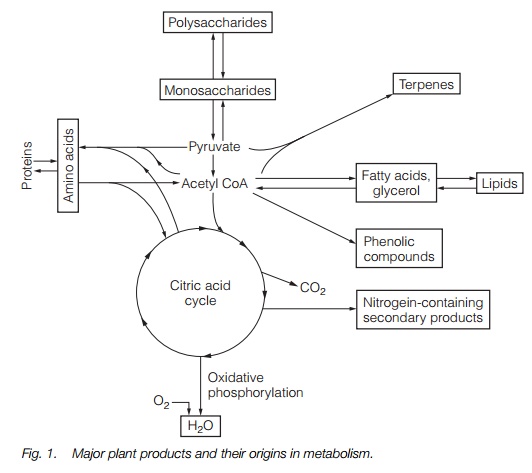
Acetyl CoA, pyruvate and citric acid cycle intermediates are the starting point for the production of amino acids, lipids, polysaccharides and secondary products in plant metabolism.
Amino acid biosynthesis
Production of amino acids is linked to the assimilation of nitrogen by the plant. Nitrate is converted to ammonium by nitrate reductase and ammonium is then incorporated into glutamine and glutamate, either by the glutamine synthase-glutamate synthase (GS-GOGAT) pathway or by glutamate dehydrogenase (GDH). Ammonium is toxic and is converted to organic nitrogen compounds in the root. Nitrate may be converted to ammonium in the roots, or is carried to shoots and leaves and either stored in the vacuole or converted to ammonium. Other amino compounds are formed by transamination reactions.
Lipid biosynthesis
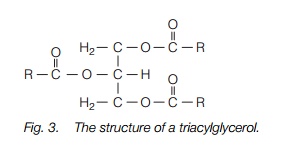
Plants synthesize a wide variety of lipids, including membrane lipids, cuticular waxes and seed storage lipids (mostly triacylglycerols). Synthesis of glycerolipids occurs in two stages: addition of the fatty acid chains to glycerol-3-phosphate and addition of a head group. Triacylglycerols consist of a glycerol to which three fatty acid chains have been added and are synthesized in the endoplasmic reticulum. They are stored in oil bodies, small lipid droplets with a surrounding coating of lipid and protein.
Sucrose, polysaccharides and starch
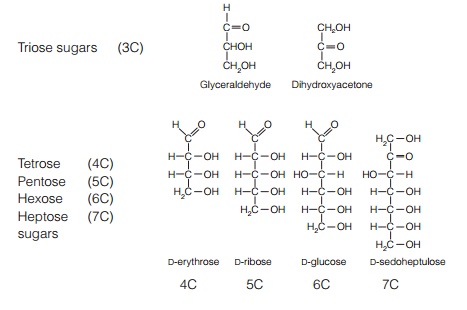
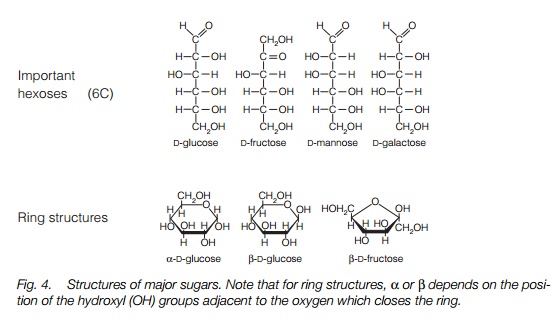
Plant cells produce monosaccharides, from 3-carbon trioses to 6-carbon hexoses. Sucrose is a disaccharide, made of glucose and fructose. Starch is a polysaccharide made up of a(1–4) and a(1–6) branched D-glucose residues. Cellulose is made up of a-D-glucose. Sucrose is synthesized in the cytoplasm either by sucrose phosphate synthase and sucrose phosphate phosphatase or by sucrose synthase.
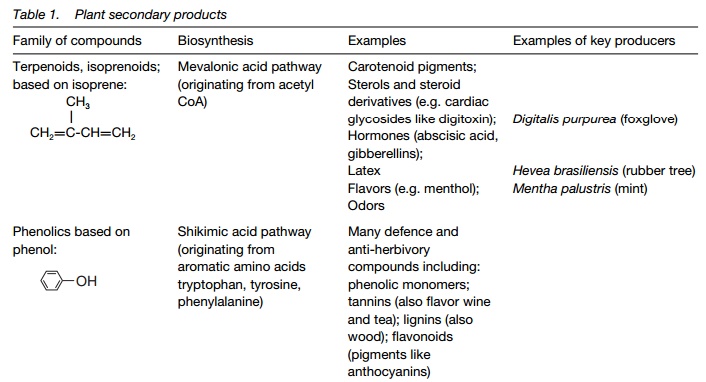
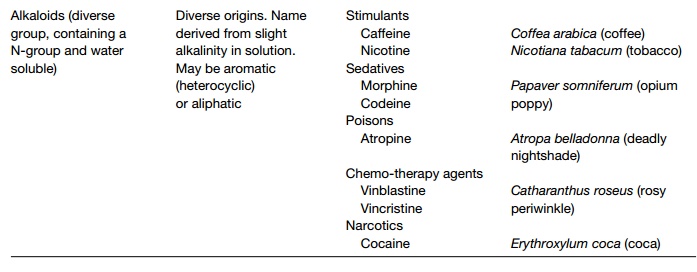
Plant secondary products
A secondary product is one that is not involved in primary metabolism. They are generally produced in specialized tissues, with highly developed multi-enzyme pathways for their production. Plant secondary products include alkaloids, terpenoids and phenolic compounds. Many are involved in plant defenses against herbivory or fungal pathogens; others are of economic importance as medicinal and industrial compounds.
Role of the citric acid cycle
Fig. 1 illustrates that pyruvate, acetyl CoA and the citric acid cycle are central in the production of many compounds. The activity of each of the pathways depends on substrate availability (usually monosaccharide) and on the tissue. As the pathways are often complex and involve many enzymes, initiating secondary product metabolism involves the developmental activation of many genes. Such gene expression and the production of secondary metabolites may be enhanced by stress, wounding, pathogen attack and herbivory.
Amino acid biosynthesis
Production of amino acids and other nitrogen-containing compounds is directly linked to the assimilation of nitrogen by the plant. Nitrogen is taken up as either nitrate or ammonium, the nitrate being converted by nitrate reductase into
ammonium. Ammonium is incorporated into glutamine and glutamate, either by the glutamine synthase-glutamate synthase (GS-GOGAT)pathway or by glutamate dehydrogenase (GDH) . Ammonium is toxic and is converted to organic nitrogen compounds in the root. Nitrate is converted to ammonium in the roots, or carried to shoots and leaves and either stored in the vacuole or converted to ammonium there.
Roots have glutamine synthase (GS) in cytosol and plastids. Root plastids also have glutamate synthase that uses NADH as electron donor (NADHGOGAT). If a root is supplied with nitrate, expression of ferredoxin-dependent (Fd)-GOGAT in plastids is induced.
Shoots have GS in the cytosol and chloroplasts. The chloroplast form takes ammonium generated by photorespiration, preventing it becoming toxic. Shoots and leaves express Fd-GOGAT in chloroplasts. Chloroplasts of shoots and leaves also contain GDH which synthesizes glutamate from ammonium and 2-oxoglutarate.
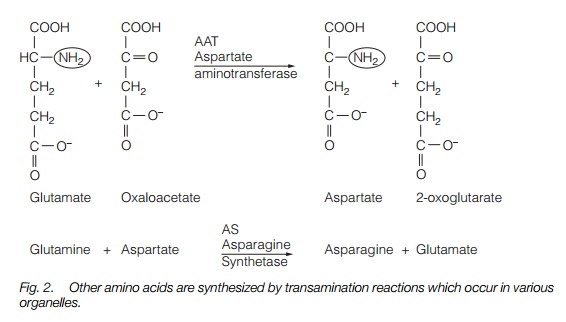
Transamination reactions involve the transfer of an amino group from one compound to another. For instance, asparagine synthase (AS; Fig. 2) converts aspartate and glutamine to asparagine and glutamate. Asparagine contains a high quantity of nitrogen (2N) for each carbon (4C) present, compared with glutamate (1N:5C) or glutamine (2N:5C). Expression of the AS gene is repressed (inhibited) by high light and high carbohydrate production, so the plant makes nitrogen compounds with low quantities of nitrogen. When light is low and carbohydrate is scarce, carbon is conserved by the synthesis of compounds with a much higher nitrogen content.
Lipid biosynthesis
Plants synthesize a wide variety of lipids, including: membrane lipids, in which long-chain fatty acids, commonly 16–18 carbons in length, are esterified to glycerol; the waxes of the cuticle and suberin of the endodermis; and the storage lipids of seeds (most commonly triacylglycerols).
Plants use acetyl CoA as the basic building block for assembling long-chainfatty acids. Acetyl CoA is produced by pyruvate dehydrogenase present both in plastids and mitochondria. In the chloroplast, pyruvate is produced from 3-phosphoglycerate from the Calvin cycle; in non-photosynthetic tissue, it originates from glycolysis. Synthesis of glycerolipids occurs in two stages: addition of the fatty acid chains to glycerol-3-phosphate and the addition of a head group. Glycerol-3-phosphate is derived in stages originating from glyceraldehyde- 3-phosphate. In photosynthetic tissue, the chloroplast is the chief source of fatty acids, which may be used directly in the chloroplast or exported to the cytoplasm. Plant cells can synthesizeglycerolipids at the endoplasmic reticulum (ER) and in mitochondria as well as in plastids; all three of these organelles are believed to be inter-connected and lipids traffic between them and to the other cellular membranes.
Seeds make triacylglycerols, which are storage oils, and frequently very important human foods. Up to 60% of the dry weight of a seed may be in the form of storage oils. Triacylglycerols consist of a glycerol to which three fatty acid chains have been added (Fig. 3). They are stored in oil bodies, small lipid droplets with a surrounding coating of lipid and protein. The oil is first synthesized in the ER.
Sucrose, polysaccharides and starch
Plant cells produce a range of monosaccharides, from 3-carbon triose sugars such as dihydroxyacetone and glyceraldehyde to 6-carbon hexoses such as D-glucose, D-fructose, D-mannose and D-galactose (Fig. 4). Sucrose is a disaccharide, made of glucose and fructose. Polysaccharides are large polymers of these monosaccharides. By far the most important of these as a storage product is starch, made up of α-(1–4) and α-(1–6) branched D-glucose residues (Fig. 4). Cellulose, a major structural component of cell walls, is another polysaccharide, made up of β-D glucose (Fig. 4).
Sucrose synthesis occurs in the cytoplasm by one of two routes as a result of the activity of two enzymes, sucrose phosphate synthase and sucrose phosphatephosphatase:

Cellulose is synthesized by cellulose synthase, an enzyme located in rosettes in the plasma membrane . Other polysaccharide complexes inserted in the cell wall are synthesized in the Golgi apparatus and secreted in vesicles, which fuse with the plasma membrane.
Regulating both the rates of sucrose and starch synthesis is important. If too much sucrose is produced, the chloroplast becomes depleted in the intermediates of the citric acid cycle, and photosynthesis will be inhibited. If too little starch is produced, the cell will not have sufficient reserves of carbohydrate for respiration during the night. Photosynthetic tissues also export assimilated carbon to other tissues and so regulation of the whole process must allow for this.
Plant secondary products
Plant secondary products are compounds generated by secondary pathways and not from primary metabolism. Many are toxic or give the plant anunpleasant taste and it is likely they give a selective advantage as anti-herbivory agents. Numerous plant secondary products have been used over many hundreds of years for a wide array of purposes (Topics N2 to N4). They are generally produced in specialized tissues, with highly developed multi-enzyme pathways for their production. Table 1 summarizes major secondary products, with their origins and uses.