Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Fishes as social animals: aggregation, aggression, and cooperation
Aggregations Shoaling of Fishes
Aggregations
Shoaling
The most obvious form of social behavior in fishes is the formation of groups, either unorganized shoals or organized, polarized schools (Fig. 22.6). By convention, some social attraction among individuals is required for a group to be considered a shoal or a school, whereas fish that are mutually attracted to food or other resources are anaggregation (e.g., Freeman & Grossman 1992). Shoals involve social attraction, coordination, and numbers. Two fish are not a shoal because one fish often leads and the other follows. However, when three or more fish co-occur, each fish reacts to the movements of all adjacent fish. The group becomes the leader and the fish become the followers. Operationally defined, a shoal is a group of three or more fish in which each member constantly adjusts its speed and direction in response to other group members; if the behavior is highly synchronized and fish swim parallel, the group is a school (Partridge 1982).
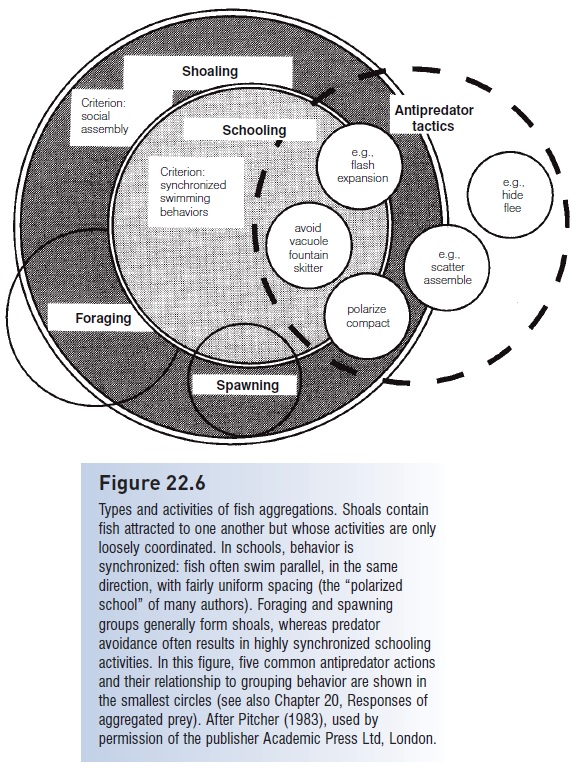
Figure 22.6
Types and activities of fish aggregations. Shoals contain fish attracted to one another but whose activities are only loosely coordinated. In schools, behavior is synchronized: fish often swim parallel, in the same direction, with fairly uniform spacing (the “polarized school” of many authors). Foraging and spawning groups generally form shoals, whereas predator avoidance often results in highly synchronized schooling activities. In this figure, five common antipredator actions and their relationship to grouping behavior are shown in the smallest circles. After Pitcher (1983), used by permission of the publisher Academic Press Ltd, London.
As many as half of all fish species may form aggregations at some time in their life. Aggregations can serve several purposes concurrently (see Parrish & Hamner 1997). The chief functions are to reduce the success of predators, increase foraging success, synchronize breeding behavior, and increase hydrodynamic effi ciency. Some species shoal throughout their lives (e.g., many herrings, anchovies, minnows, silversides), others only as juveniles (Bowfin, plotosid catfishes, surgeonfishes, pufferfishes). Some species aggregate when young, disband as juveniles or adults, and reaggregate to spawn, either in groups or as pairs (many salmonids, seabasses). Foraging aggregations may turn into breeding aggregations as fishes migrate to traditional spawning locations and are joined by members of other aggregations (Yellow Perch, grunts, rabbitfishes). Normally solitary adults may congregate during the winter and such aggregations probably remain together through a spring spawning season (e.g., carp).
Within a species, schooling tendency may change with predation intensity. Guppies in predator-dense habitats school throughout their lives, but only juveniles school where predators are rare. European Minnows that co-occur with predators inherit a stronger schooling tendency than do minnows without predators (Magurran 1990). The intense predation pressure that small fishes experience and the prevalence of shoaling behavior particularly among juveniles and small species attests to the antipredator func
Regardless of function, most fish shoals are relatively unstable. Few fishes, at least in inshore locales, maintain their groups through an entire 24 h period. Many shoals form each morning, disband at night, and reform the following morning but with different individuals. In fact, available evidence indicates little shoal fi delity in most fishes: dace, minnows, killifish, Yellow Perch, surgeonfishes, parrotfishes, and Bluegill Sunfish join and leave foraging shoals frequently (Freeman & Grossman 1992; Hoare et al. 2000). Climbing Perch, Anabas testudineus (Anabantidae), even show a preference for shoaling with unfamiliar individuals, especially if unfamiliar fish are part of a larger group (Binoy & Thomas 2004), and European Minnows, Phoxinus phoxinus, will prefer shoals of unfamiliar fish when the unfamiliar fish are in a significantly larger shoal (Barber & Wright 2001). However, advances in molecular genetic techniques are revealing a degree of permanence in the form of stable kin groups in some wild fish schools (e.g., Brook Char, Salvelinus fontinalis; Fraser et al. 2005; EurasianPerch, Perca fluviatilis, Behrmann-Godel et al. 2006).
Fidelity may also be strong in nocturnal fishes that reaggregate each dawn and form daytime resting schools at fixed refuge locales (e.g., squirrelfishes, grunts, copper sweepers, some bullhead catfishes; Hobson 1973; Helfman 1993). In some nocturnal species, relatively complex social structure and interactions develop that rival the societies of birds and mammals. Migratory schools (e.g., large tunas, Bluefishes) may also show strong group fi delity, but definitive information is lacking.
Fishes space themselves fairly regularly within fish schools (e.g., Partridge et al. 1983; Abrahams & Colgan 1985). A perfect crystal lattice is the theoretical ideal spatial distribution for school members: neighbors should be 0.3–0.4 body lengths apart, 5 body lengths behind and centered between preceding fish, with neighbors beating their tails in antiphase (opposite directions). Fish could gain a 65% energy saving from the wakes and vortices generated by fish around them (Weihs 1975). However, few if any groups achieve the proposed ideal lattice structure (Pitcher & Parrish 1993; Parrish & Turchin 1997). Regardless, considerable energy savings may accrue in a school because the fish are doing more than simply “drafting” in a region of reduced flow created by the fish ahead of them (Liao et al. 2003). Some fishes contract only anterior body muscles, allowing a passive wave of undulation to propagate posteriorly along the body, much as waves pass down a flag in the wind. A fish thus progressively slaloms between the vortices shed by the fish swimming ahead of it, matching its swimming movements in phase with oncoming drag vortices, rather than expending energy pushing off the vortices (similar energy savings are experienced by fish undulating behind a rock or other object in flowing water).
Earlier authors had proposed that a hydrodynamic advantage may also develop through drag reduction whenone fish swims through the mucus produced by fish ahead of it in a school (Breder 1976). However, insuffi cient mucous is produced, even in schools of a billion fish, to affect drag significantly (Parrish & Kroen 1988). Predation also disrupts spacing because individuals under attack should attempt to place schoolmates between themselves and a predator (the selfish herd phenomenon; Hamilton 1971). Again, however, experimental tests call into question whether central locations are in fact safer (Parrish 1989b). Obviously, many factors (size, sensory input to the lateral line, visibility of neighbors, swimming speed, species composition, vulnerability to and behavior of predators, social status) contribute to the exact (and variable) structure of schools.
Colonial fishes form essentially stationary aggregations. Colonies may exist for breeding, as when male sunfishes and cichlids and female triggerfishes aggregate and construct nests or set up display sites. Some damselfishes set up contiguous territories in suitable habitat patches on a coral reef. Three-spot and Bicolor damselfishes will be found in areas of a few square meters, even though adjacent, similar reef areas contain no such fishes (Schmale 1981). Garden eels (Congridae) occupy small burrows a few centimeters apart on sandy regions of coral reefs (Fig. 22.7). Jawfishes (Opisthognathidae), another burrowing coral reef form, tend to form colonies of two to nine individuals on rubble-strewn sandy bottoms (Colin 1973).

Related Topics