Chapter: Biochemical Pharmacology : Introduction
What are drugs?
What are drugs?
Do drug molecules have
anything in common at all? Figure 1.1a shows the structure of the smallest drug
- molecular (or, more precisely, atomic) weight 6 Da.
On the other end of the
scale, we have a rather large molecules – proteins. Shown is the structure of
tissue plas-minogen activator (t-PA; Figure 1.1b). t-PA is a human pro-tein.
Its tissue concentration is very low, but by means of recombinant expression in
cell culture it can be obtained in clinically useful amounts. t-PA is now the
`gold stan-dard' in the thrombolytic therapy of brain and myocardial
infarctions.
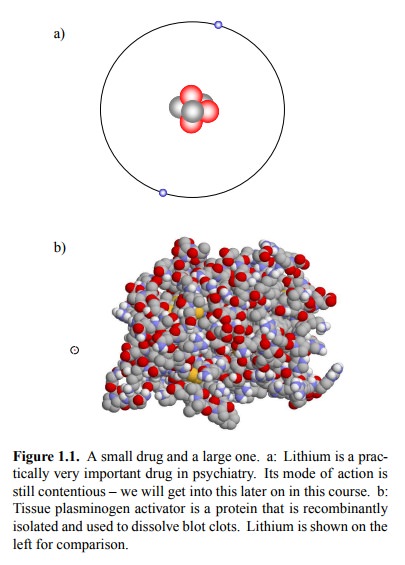
The molecular weight of t-PA
is about 70 kDa. Few drug molecules (among them the increasingly popular
bo-tulinum toxin) are bigger than t-PA.
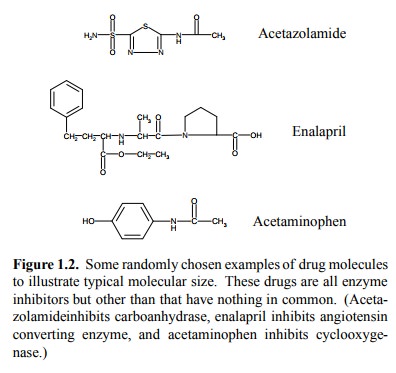
More typical sizes of drug
molecules are shown in Figure 1.2. Most practically useful drugs are organic
molecules, with as molecular weight of roughly 200 to 2000, mostly below 1000.
Interestingly, this also applies to many natural poisons (although on average
they are probably somewhat larger). Are there reasons for this?
Reasons for an upper limit
include:
1. Most drugs are chemically synthesized (or at
least mod-ified, e.g. the penicillins) – the larger the molecules, the more
difficult the synthesis, and the lower the yield will be.
2. Drugs need to reach their targets in the body,
which means they need to be able to cross membrane barriers by diffusion.
Diffusion becomes increasingly difficult with size.
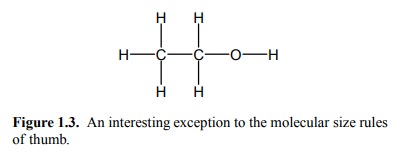
One argument for a lower
limit may be the specificity that is required – drugs need to act selectively
on their target molecules in order to be clinically useful. There are numer-ous
examples of low-molecular weight poisons – proba-bly the better part of the
periodic table is poisonous. There are, however, interesting exceptions to
these molecular size rules of thumb. One is lithium; another popular example is
shown in Figure 1.3.
Related Topics