Chapter: Biochemical Pharmacology : Introduction
Drugs and drug target molecules
Drugs and drug target
molecules
Drugs need to bind to target
molecules. Is there anything remarkable about this statement at all? Well, two
things:
1. It is a surprisingly recent insight – only
about 100 years old. (OK, so that is relative – long ago for you, but I'm
nearly there.)
2. It is not generally true.

The idea of defined receptor
molecules for drugs or poisons was conceived by Paul Ehrlich (Figure 1.4).
Ehrlich worked on a variety of microbes and microbial toxins. He observed that many dyes used to stain specific
structures in micro-bial cells in microscopic examinations also exerted toxic
effects on the microbes. This observation inspired him to systematically try
every new dye he could get hold of (and new dyes were a big thing in the late
19th century!) on his microbes. Although not trained as a chemist
himself, he managed to synthesize the first effective antibacterial drug – an
organic mercury compound dubbed `Salvarsan' that was clinically used to treat
syphilis for several decades, un-til penicillin became available. Ehrlich
screened 605 other compounds before settling for Salvarsan. In keeping with his
enthusiasm for colors and dyes, Ehrlich is credited with having possessed one
of the most colorful lab coats of all times (he also had one of the most
paper-jammed offices ever). His Nobel lecture (available on the web) is an
inter-esting read – a mix of brilliant and utterly `naive' ideas that makes it
startlingly clear how very little was known in biol-ogy and medicine only a
century ago.
So, what molecules are targets of drugs? Some typical
ex-amples are found in the human renin-angiotensin system, which is important
in the regulation of blood pressure (Fig-ure 1.5. Angiotensinogen is a plasma
protein that, like most plasma proteins, is synthesized in the liver. From this
pro-tein, the peptide angiotensin I is cleaved by the specific pro-tease renin,
which is found in the kidneys (ren
lt. = kidney). Angiotensin I, which is only weakly active as a mediator, is
cleaved further by angiotensin converting enzyme, which is present in the
plasma. This second cleavage releases an-giotensin II, which is a very powerful
vasoconstrictor. An-giotensin II acts on a G protein-coupled receptor,
amem-brane protein that is found on vascular smooth muscle cells. Through a
cascade of intracellular events, this receptor triggers contraction of the
muscle cell, which leads to con-striction of the blood vessels and an increase
of blood pres-sure).
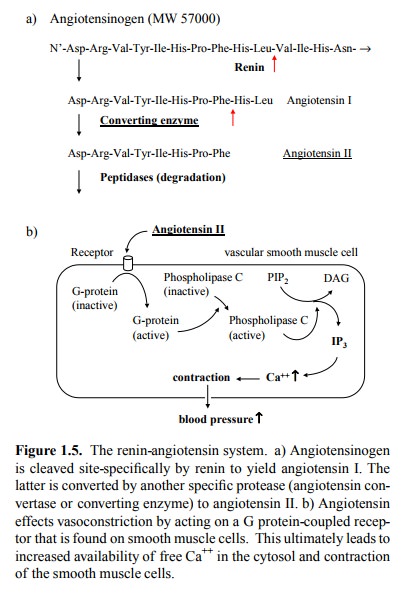
Increased activity of the renin-angiotensin
system is fre-quently observed in kidney disease, which may lead to ab-normally
high release of renin. Several points in the system are amenable to
pharmacological inhibition. The first one is renin itself, which splits a
specific bond in the angiotensino-gen polypeptide chain (Figure 1.5a). An inhibitor
of renin is remikiren (Figure 1.6a).
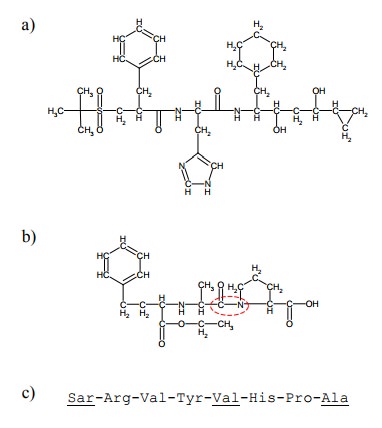
Remikiren (Figure 1.6a) is
effective but has several short-comings, such as low `bioavailability' – which
means that the drug does not efficiently get into the systemic circula-tion
after oral uptake. Of course, oral application is quite essential in the
treatment of long-term conditions such as hypertonia. A major cause of low
bioavailability of drugs is their metabolic inactivation. Drug metabolism
mostly happens in the liver (and sometimes in the intestine) and of-ten is a
major limiting factor of a drug's clinical usefulness. Remikiren contains
several peptide bonds, which likely are a target for enzymatic hydrolysis.
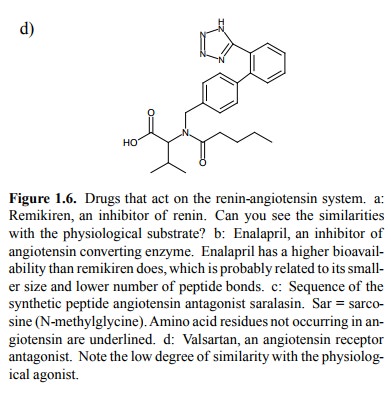
The most practically
important drugs that reduce an-giotensin activity are blockers not of renin but
of an-giotensin converting enzyme blockers, such as enalapril (Figure 1.6b).
These have a major role in the treatment of hypertonia. In contrast to
remikiren, enalapril is of smaller size and has only one peptide bond, which is
also less acces-sible than those of remikiren. These features correlate with a
bioavailability higher than that of remikiren.
Related Topics