Chapter: Biochemical Pharmacology : Introduction
Metabolism of physiological mediators and of drugs
Metabolism of physiological
mediators and of drugs
So far, we have encountered two reasons for
designing drug molecules that are structurally different from physiological
mediators:
1. Turning an agonist into an inhibitor, and
2. Increasing receptor selectivity.
Both these reasons relate directly to the
interaction of the drug molecule with its target. A third rationale for varying
the structure of the drug molecule is that most physiological mediators are
rapidly turned over in the organism, which is usually undesirable with drugs.
E.g., angiotensin lives only for a few minutes (as does saralasin); the same
applies to epinephrine and norepinephrine2. With these, one
impor-tant pathway of inactivation consists in methylation (Figure 1.10).
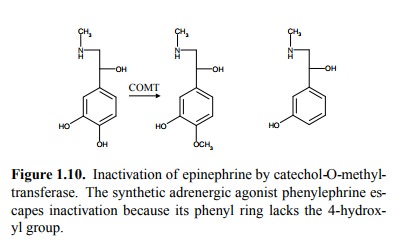
The drug phenylephrine
(Figure 1.10, right) lacks the cru-cial hydroxyl group that normally initiates
inactivation of epinephrine and therefore persists for hours rather than
minutes in the organism, making it more practically useful in pharmacotherapy
(`take this twice daily with the meal'). Its lower intrinsic affinity to the
receptor (about 100fold lower than that of adrenaline) can be offset by
increasing the absolute amount applied.
In practical pharmacotherapy, a drug's
metabolism and elimination are of equal importance as its specific mecha-nism
of action. There are several reasons for this:
1. Drugs may be extensively metabolized in the
liver. Since all orally applied drugs are passed through the liv-er before
reaching the systemic circulation, this can lead to impractically low effective
levels at the relevant target site. Example: Remikiren (above).
2. Sometimes, the metabolic products are more
active than the parent drug, or they may have poisonous effects that were not
observed with the parent compound itself3.
3. Diseases – or concomitant use of other drugs –
may significantly change the rate of metabolism and thereby change the
bioavailability of the drug, leading to loss of desired effects or unacceptably
severe side effects.
In the foregoing, we have
seen several examples of one frequently used approach to drug development: The
struc-ture of a physiological mediator is used as a starting point; a large
number of variants are synthesized, and from the pool of variants those with
the desired agonistic or antag-onistic properties are `screened' using
appropriate in vitro assays and
animal experiments. This approach does not al-ways work. Below are some
examples of other successful approaches to drug development. You will note that
some of these are not completely general either.
Related Topics