Chapter: Health Management in Aquaculture: Immunological and molecular biology techniques in disease diagnosis
Western Blotting - Aquaculture Immunological techniques in disease diagnosis
Western Blotting
Western blotting is a rapid and sensitive assay for the detection and characterization of proteins. The technique allows one to identify particular proteins by utilizing the specificity inherent in antigen-antibody recognition. This tech-nique is powerful, since it combines electrophoretic separation of proteins, gly-coproteins and lipopolysaccharides with immunological identification. Once such antigens have been detected, they can be further characterized by Western blotting. Both techniques can utilize either polyclonal or monoclonal anti-bodies.
Initially, a sample is subjected to electrophoresis to separate antigens according to their charge and size, or size alone. A second electrophoretic step transfers the antigens from the gel to an immobilizing surface, such as nitrocellulose
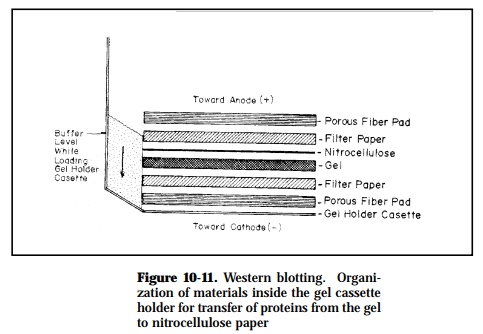
paper where they are bound irreversibly (Figure 10-11). After this transfer, the paper is blocked with 3% gelatin in PBS to prevent nonspecific binding of anti-body and probed with a specific enzyme-conjugated antibody (horseradish peroxidase-anti-immunoglobulin conjugate). A chromogenic substrate is then added to determine which electrophoretic band is bound by the antibody.
The technique is useful for a number of purposes including characterization of unknown antigens or antibody specificities, confirmation of the presence of bacterial antigens in sera or tissues and detection of seropositive individuals which have been exposed to a pathogen. The primary advantage of Western blotting, as opposed to other immunoassays, is the high degree of specificity in resolving distinct antigens. However, there are two disadvantages: first, it is mainly a qualitative assay and quantification of antibody or antigen is difficult; and second, if the antigen sample must be denatured (such as in SDS-PAGE), antigenic activity may be reduced or destroyed. An initial consideration for successful Western blotting is the empirical determination of the optimal per-centage of acrylamide for resolution of the antigens of interest. Next, it must be determined whether the antigens are capable of binding to the nitrocellu-lose. Finally, the antigen detection procedure must be highly specific and sensitive.
The technique can be used to detect as little as 1 ng of a protein antigen that has been previously separated under denaturing conditions by SDS-PAGE, pro-vided that an antibody that recognizes the denatured form of the protein is available. The entire procedure can be completed in 1 to 2 days, depending on transfer time and type of gel.
Western blotting has been useful for characterizing the specificities of polyclonal antisera (rabbit and salmonid) and monoclonal antibodies to extra-cellular and cell surface antigens of Renibacterium salmoninarum (Wiens and Kaattari, 1989). It has also been used in the detection of yellowhead virus and white spot syndrome virus in penaeid shrimp (Nadala et al., 1997; Magbanua et al., 2000).
Related Topics