Chapter: Health Management in Aquaculture: Immunological and molecular biology techniques in disease diagnosis
Fluorescent Antibody Technique (FAT) - Aquaculture Immunological techniques in disease diagnosis
Fluorescent Antibody Technique (FAT)
Searching for bacteria in fish tissue is easier than defining viral infections by FAT. In most cases when antisera against viral fish pathogens are used, the fish tissues suspected of being infected are processed, filtered and placed in culture on fish cell lines; the purpose being to increase the amount of virus.
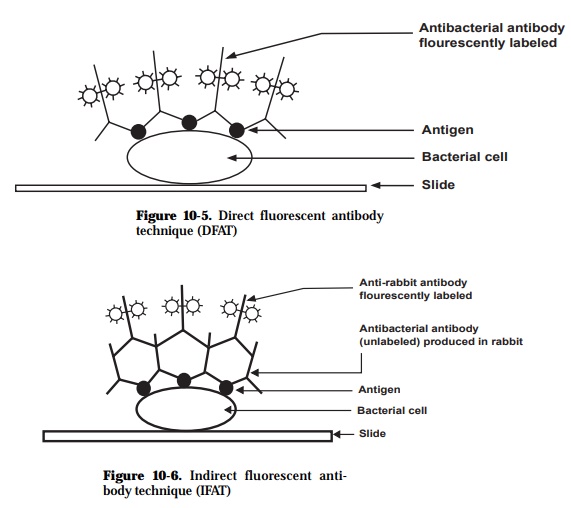
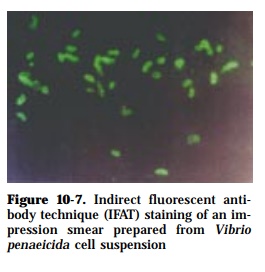
The FAT makes use of the feature that some dyes (fluorescein isothiocyanate, rhodamine isothiocyanate, Texas red) fluoresce when viewed under ultraviolet (UV) light. These dyes can be used as tags, conjugated to target antibody mol-ecules, and when the antibody complexes with the specific antigen, the com-plex is “lighted”. A major advantage of being able to visualize the antigen-antibody reaction on the pathogen gives the pathologist confidence in diagno-sis. This technique generally detects surface-associated antigens. Slide for-mats, with organisms fixed directly to the slides, are used for bacteria. After fixation, antigen is incubated with specific antibody. This may be labeled di-rectly with fluorescein to allow visualization of antibody (direct FAT) (Figure 10-5) or, alternatively, a fluorescein-labeled antibody conjugate is added in a second stage to detect bound antibody (indirect FAT) (Figures 10-6 and 10-7). Slides are then examined with an ultraviolet microscope, and the intensity of fluorescence, relative to appropriate controls, is scored in an arbitrary scale from -, +, to +++++. Typical assays can be performed in 2-3 h.
Immunofluorescence is widely used for the detection of antigen or antibody in fish, typing and identification of a range of microorganisms. FAT has been used to detect antibodies to Aeromonas liquifaciens in fish (Lewis and Savage, 1972). Rapid FAT diagnosis has been developed to detect Pseudotuberculosis in yellowtail (Kitao and Kimura, 1974), Renibacterium salmoninarum in salmo-nids (Bullock et al., 1980) and Vibrio penaeicida in kuruma prawn (de la Peña et al., 1992). FAT has also been developed for rapid diagnosis of infectious hematopoeitic necrosis virus (IHNV) (LaPatra et al., 1989) and red sea bream iridovirus (Nakajima et al., 1995) in fish.
Related Topics