Chapter: Biochemical Pharmacology : The ionic basis of cell excitation
Voltage-gated cation channels and the action potential
Voltage-gated cation channels and the action potential
While transporters and leak
channels shape the resting po-tential, the action potential is the fiefdom of
the gated channels. Voltage-gated channels, in particular, are important for
the spreading of action potentials over the surface of an entire cell. In most
excitable cells, there are high numbers of voltage-gated channels for both
sodium and potassium (and, in fact, several subtypes of both). In addition,
voltage-gated Ca++ channels prominently occur in heart and smooth muscle
cells.
In the resting state, the
voltage-gated channels are closed. As stated before, the negative-inside
potential at rest is due to the occurrence of K+ leak channels
(Figure 4.5a). The voltage-gated channels enter into the picture when a change
occurs in the surrounding electrical field. The channel pro-teins possess
mechanically flexible domains that carry an electrical charge, and that will
thus change conformation in response to Coulomb forces. If the electrical field
is re-versed artificially using microelectrodes, the gate of the sodium channel
`swings' open (Figure 4.5b), and all of a sudden the permeability of the cell
membrane for sodium will exceed that for potassium. According to the Goldman
equation, this will lead to the re-orientation of the mem-brane potential,
because the distribution of Na+ ions is op-posite to that of K+.
This reversal of the membrane is called depolarization. Therefore, the membrane
responds to a lim-ited extrinsic depolarization with a much more vigorous
de-polarization of its own. This is what constitutes membrane excitation.
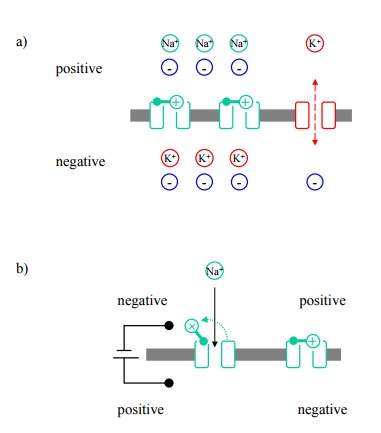
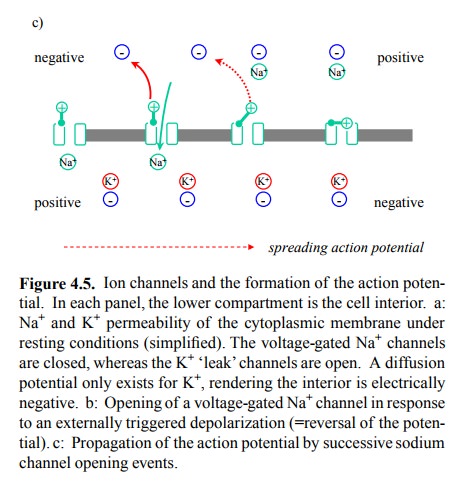
Figure 4.5. Ion channels and the formation of the action poten-tial. In each panel, the lower compartment is the cell interior. a: Na+ and K+ permeability of the cytoplasmic membrane under resting conditions (simplified). The voltage-gated Na + channels are closed, whereas the K+ `leak' channels are open. A diffusion potential only exists for K+, rendering the interior is electrically negative. b: Opening of a voltage-gated Na+ channel in response to an externally triggered depolarization (=reversal of the poten-tial). c: Propagation of the action potential by successive sodium channel opening events.
A crucial aspect of this excitation is that it
will spread along the entire expanse of the membrane. The initial, trigger-ing
depolarization is usually a localized event; the opening of the sodium channels
close by then provides the trigger for channel opening and depolarization in
the adjoining re-gions in turn. This self-sustained, spreading depolarization
is called the action potential (Figure 4.5c).
Although the simplified
cartoons suggest otherwise, the membrane potential does not actually have to be
reversed for the sodium channels to open. As soon as the externally triggered
depolarization reaches a threshold value of about -55mV (Figure 4.6b), the
sodium channels will start to open, and the action potential will be triggered,
causing the depolarization to spontaneously go up to about +50-60 mV (Figure
4.6a).
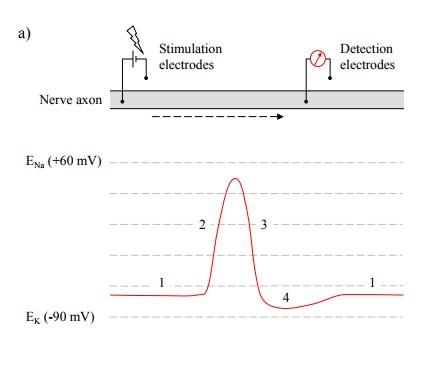
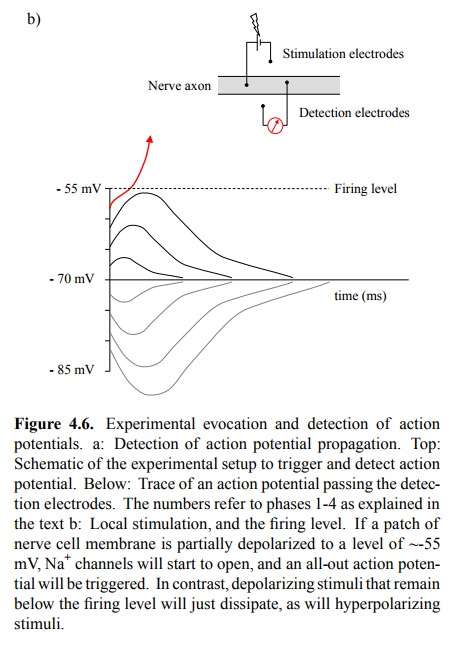
Figure 4.6a shows the trace of an action
potential traveling down a nerve fiber, recorded at some distance downstream of
the stimulating electrode. It consists of a brief depolarization spike
(duration ~1-3 msec) that approaches but does not quite reach the Na+
equilibrium potential, followed by a slightly longer lasting depression below
the level of the resting potential. Altogether, we can distinguish four phases
in the potential curve (see numbers in Figure 4.6a), which can be explained as
follows:
·
Phase 1
is the resting potential, maintained close to the K+ equilibrium
potential by the K+ leak channels.
·
The
vigorous rise of the action potential is caused by the opening of voltage-gated
Na+ channels. With the Na+ permeability now exceeding the
K+ permeability, the membrane potential is shifted toward the Na+
equilibri-um potential.
·
The
equally rapid decline is due to the Na+ channels beginning to close,
and voltage-gated K+ channels be-ginning to open. The combined
permeabilities of the voltage-gated and the leak K+ channels exceed
the de clining Na+ permeability and pull the potential back to-ward
the K+ equilibrium potential.
· The transient hyperpolarization is due to open
voltage-gated K+ channels lingering after the Na+
channels have already closed.
·
Like the
sodium channels, the voltage-gated potassium channels will finally inactivate.
This will revert the membrane potential back to normal.
Thus, two effects are
responsible for the limited duration of the action potential (essential if we
don't want to throw away the brain cell after single use, kind of wasteful):
1. The inactivation of the sodium channels, which
after having stayed open for just a few milliseconds will close despite
persisting membrane depolarization;
2. The opening of the voltage-gated potassium
channels, which will pull the membrane potential back into the direction of the
K+ equilibrium potential.
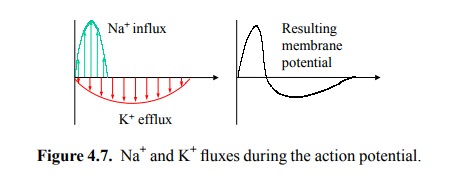
While the workings of the
sodium and the potassium chan-nels are closely similar, the sodium channels are
faster at both opening and closing. This gives the action potential its shape
(Figure 4.7).
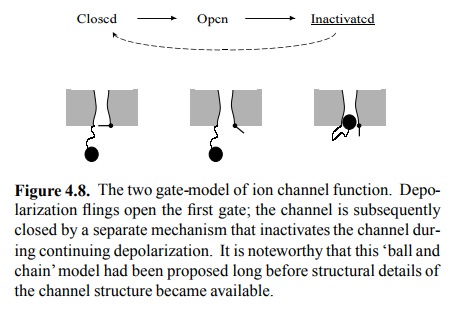
If depolarization is
responsible for opening the channel gate, it can not easily be blamed for its
closing (cf. Figure 4.5b). To account for the closing event, we must therefore
postulate a separate mechanism (Figure 4.8). The struc-ture of the
voltage-gated potassium channel is more or less known (Figure 4.9), and the
gate mechanisms for both opening and inactivation can actually be located.
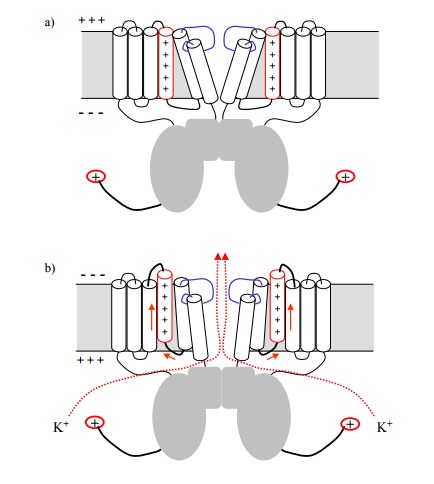
The channel consists of four
protein subunits, each of which contributes 6 helices to transmembrane part of
the channel. In addition, a sizeable chunk of protein protrudes into the
cytosol. The trigger of the opening gate is located on one of the 6
transmembrane helices, which carries sev-eral positive charges (an unusual feature
for transmembrane helices, which typically have no charges at all). The charge
will cause the helix to move when the electrical field in its vicinity is
changed, i.e. when the membrane is depolarized. This movement will cause
adjacent helices to move in turn and thus open the gate (Figure 4.9b).
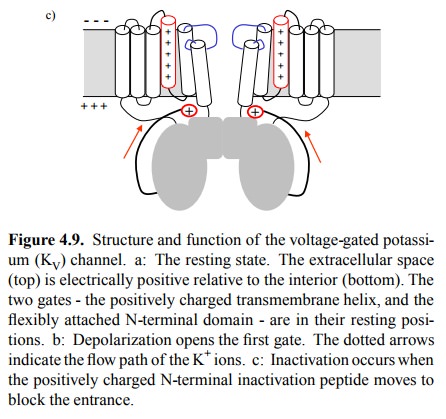
The second gate, which is
responsible for inactivation, conforms very much to the ball-and-chain
mechanism de scribed in Figure 4.8. It consists in a positively charged,
flexibly attached N-terminal domain, which likewise under-goes a movement in
the changed electrical field and simply plugs the pathway that is transiently
opened for the K+ ions (Figure 4.9c).
Related Topics