Chapter: Biochemical Pharmacology : The ionic basis of cell excitation
Ion gradients across the cell plasma membrane
Ion gradients across the cell
plasma membrane
All membrane potentials
depend on the existence of ion gradients across the membrane in question. The
major ion species that shape the form of both resting potentials and action
potentials are K+, Na+, Ca++, and Cl-.
The ion gradi-ents result from the activities of three types of membrane
proteins:
1.Ion pumps. These proteins
use metabolic energy in the form of ATP to transport ions against their
concentra tion gradients. Quantitatively the most important ion pump is Na+/K+-ATP'-ase
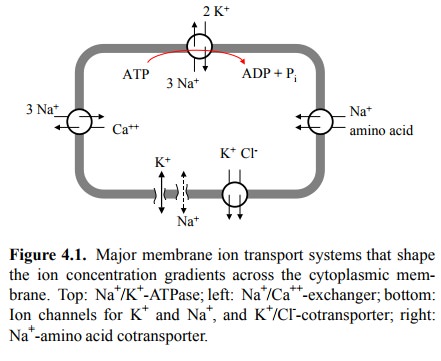
(Figure 4.1), which transports both sodium and potassium against their
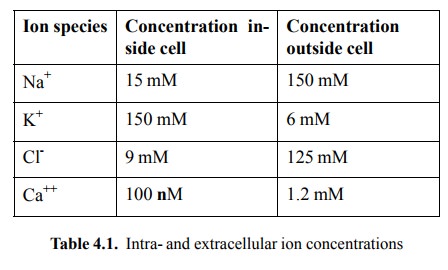
respective gra-dients (table 4.1). In addition, various types of calcium pumps
are found in the cytoplasmic, ER and mitochon-drial membranes; the direction of
Ca++ transport is al-ways from the cytosol to the other compartment.
2. Exchange- and co-transporters. These link the
gradients of different ion species to one another, so that gradients can be
established for ions for which specific pumps do not exist (or have
insufficient capacity). Important ex-amples are the sodium/calcium exchanger
and the potas-sium/chloride co-transporter in the cytoplasmic mem-brane (Figure
4.1).
3. Ion channels. These proteins simply facilitate
the diffu-sion of ions downhill their concentration gradients, i.e. they tend
to dissipate the concentration gradients estab-lished by the transporters.
Most, but not all channels
can switch between open and closed states. Switching can be accomplished by a
ligand binding to the channel or by changes in the surrounding electrical
field. Therefore, we have the following major functional groups of ion
channels:
1. Ligand-gated channels, which may either open or
close
in response to ligand
binding. Important examples are the nicotinic acetylcholine receptor, which
allows Na+ into the cell in response to acetylcholine, and the
sul-fonylurea receptor-associated Kir channel, which ceases to
permit efflux of K + in response to ATP.
2. Voltage-gated channels. The voltage-gated
channels for K+, Na+ and Ca++ are all involved
in cell excitability.
3. `Leak' channels, which seem to be fairly
simple-mind-ed and just continuously permit flux of the cognate ion. The most
important ones are those for K+; they are re-sponsible for the fact
that K+ permeability dominates the resting potential.
The continuous flux of ions
through leak channels (and ex-change transporters) requires continuous
operation of the ion pumps. Therefore, a sizeable fraction of our metabolic
energy is expended just to keep up the ion gradients across our cell membranes.
The major ion transport
processes that are responsible for maintaining the ion gradients and the
resting potential across the cytoplasmic membrane are summarized in Figure 4.1:
1. Na+/K+-ATPase (top)
exchanges 3 Na+ with 2 K+ ions for each molecule of ATP
consumed.
2. Ca++ is transported in exchange for
Na+, so that the Ca++ gradient depends in part on the Na+
gradient. In addition, Ca++ is also extruded by specific pumps (not
shown).
3. Chloride is expelled from the cytosol by
co-transport with K+. The chloride gradient therefore is dependent
on1and sustained by the K+ gradient.
4. The leak channels cause the permeability of K+
to be higher than for any other ion species, so that the resting potential is
kept close to the K+ equilibrium potential (see below).
Note that the extra Na+
ion that is extruded by Na+/K+-ATP'ase is not the immediate cause of the
negative-inside membrane potential. Instead, extra sodium ions are allowed back
in during active transport of metabo-lites such as amino acids and (with some
cells) glucose, and in exchange for Ca++; any remaining
intracellular cation deficit would be balanced by K + flowing back
in across its own channels.
Related Topics