Chapter: Obstetrics and Gynecology: Infectious Diseases in Pregnancy
Varicella - Infectious Diseases in Pregnancy
VARICELLA
Congenital
varicella (chicken pox) infection can be seri-ous, but it is
very uncommon due to high rates of immunity in women of reproductive age. Risk
for congenital varicella syndrome (skin scarring, limb hypoplasia,
chorioretinitis, microcephaly) is limited to maternal infection occurring in
the first half of pregnancy. Most patients are immune, even if they or their
families do not recall the patient having been infected. A pregnant patient
exposed to varicella can have serologic testing (IgM and IgG), and can be given
varicella zoster immune globulin within 72 hours of exposure to re-duce the
severity of maternal infection. A
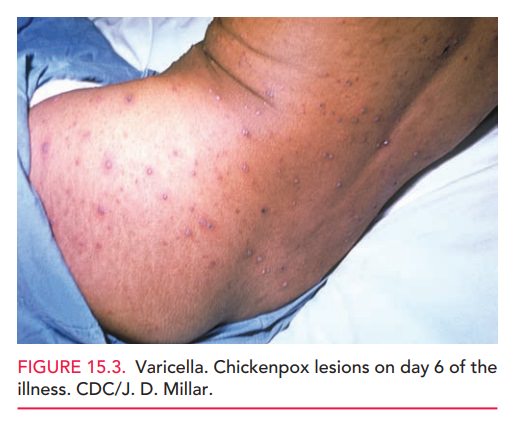
pregnant patientwho develops the characteristic varicella rash can be given
oral acyclovir within 24 hours of the rash to decrease symptoms and duration of
disease (Figure 15.3). However, maternal acy-clovir has not been shown to
decrease the rate or severity of fetal infection.
If clinical infection occurs in a
patient from 5 days prior to delivery to 2 days after delivery, neonatal
infec-tion can be severe, even deadly. Varicella zoster immune globulin is
given to infants in such situations, though pro-tection is not complete.
Severe complications of varicella including pneumo-nia and encephalitis are much more common in adults than in children. Varicella pneumonia seems to occur more frequently with varicella infection during pregnancy and is as-sociated with maternal mortality. Treatment is with intra-venous acyclovir. Effective vaccination against varicella has been available since 1995 and should be offered to suscep-tible nonpregnant women.
The
vaccine is a live attenuated virus and should be avoided in pregnancy and
within 1 month of conception; however, no adverse outcomes have been reported
if given in pregnancy.
Vaccination of susceptible
household contacts of pregnant women is safe.
Related Topics