Chapter: Biochemistry: Cell Membrane
Surface tension

Surface tension
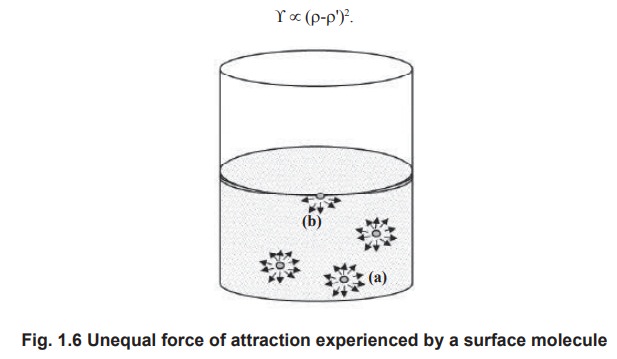
A molecule inside the liquid mass (a) is pulled
uniformly on all the sides by intermolecular forces. But a surface molecule (b)
suffers a much greater intermolecular attraction towards the interior of the
liquid than towards the vapour phase, because fewer molecules are present in
the vapour phase. The excess of inward force on the surface layer accounts for
the surface tension. Surface tension (¡) is defined as the force acting
perpendicularly inwards on the surface layer of a liquid to pull its surface
molecules towards the interior of the liquid mass.
1. Factors affecting surface tension
·
Density
- Macloed’s equation relates surface tension to the density of the liquid (r)
and that of its vapour (r’).
∝(r-r')2.
·
Temperature-
Temperature and surface tension are inversely related to each other. As the
temperature of the liquid increases, the surface tension decreases and becomes
zero at the critical temperature.
·
Solutes
- Solutes that enter the liquid raise the surface tension of the solvent, while
solutes that concentrate on the surface lower the surface tension.
2. Biological Importance
·
Emulsification
of fats by bile salts - Bile salts lower the surface tension of the fat
droplets in the duodenum, which aids in digestion and absorption of lipids.
·
Surface
tension of plasma: The surface tension of plasma is 70 dynes/cm, which is
slightly lower than that of water.
·
Hay’s
test for bile salts - The principle of surface tension is used to check the
presence of bile salts in urine. When fine sulphur powder is sprinkled on urine
containing bile salts ( as in jaundice), it sinks due to the surface tension
lowering effect of bile salts. If there are no bile salts in urine as in normal
individuals, it floats.
·
Dipalmitoyl
lecithin is a surfactant that is secreted by the lung alveoli, which reduces
the surface tension and prevents the collapse of lung alveoli during
expiration. Certain pre-mature infants have low levels of this surfactant
leading to acute respiratory distress.
Related Topics