Chapter: Paediatrics: Neurology
Subdural haemorrhage in a child under 2 years
Subdural haemorrhage in a child under 2 years
Children under 2yrs presenting
with subdural haemorrhage (SDH) are an important cause of morbidity and
mortality. A significant number will have been caused by purposeful, inflicted,
trauma, as part of an acceleration/ deceleration injury. In the investigation
of non-accidental head injury (NAHI) it is important to differentiate inflicted
injury and other causes of SDH. This section aims to help doctors and other
staff thoroughly investi-gate the child presenting with SDH.
┬Ę This section assumes that the
child is stable clinically (airway, breathing, circulation) and relevant teams
are being contacted for further opinions.
┬Ę The investigations will take at
least a week to perform, therefore there is no ŌĆśhurryŌĆÖ to produce definitive
report/guidance for other professionals until results are available.
┬Ę Do not use the term ŌĆśshaken baby
syndromeŌĆÖ. Shaking is a possible mechanism of injury, and not a syndrome, and
should be considered in the context of other mechanisms of non-accidental head
injury.
┬Ę NAHI is a leading cause of death
and disability in children, particularly if cerebral injury is part of the
spectrum of damage. Bleeding from torn bridging veins into the subdural space
is the hallmark of non-accidental head injury. When infants who developed a SDH
after infection or neurosurgical intervention are excluded, in retrospective
studies, 24ŌĆō82% of cases with SDH were highly suggestive of abuse in different
series.
Differential diagnosis of SDH
┬Ę Trauma, traumatic labour.
┬Ę Neurosurgical complications,
cranial malformation (aneurysm, arachnoid cyst).
┬Ę Cerebral infections.
┬Ę Coagulation and haematological
disorders.
┬Ę Metabolic (glutaric aciduria,
galactosaemia).
┬Ę Biochemical disorders
(hypernatraemia).
Symptoms/signs of acute SDH
┬Ę Encephalopathy (irritability,
crying, inconsolability, unsettled behaviour, lethargy, meningism, decreased or
increased tone, seizures, impaired consciousness).
┬Ę Vomiting, poor feeding.
┬Ę Breathing abnormalities, apnoea.
┬Ę Pallor, shock.
┬Ę Tense fontanelle.
Early post-traumatic seizures
occur more frequently in inflicted than in non-inflicted head injury.
Symptoms/signs of subacute or chronic SDH
┬ĘExpanding head circumference.
┬ĘVomiting, failure to thrive.
┬ĘNeurological deficit/s.
Retinal haemorrhages (ŌĆśhaemorrhagic retinopathyŌĆÖ)
┬ĘAlthough strongly associated with
NAHI, retinal haemorrhages are not specific for the diagnosis, nor can they be
dated with precision. In NAHI haemorrhagic retinopathy can typically affect all
retinal layers. It shows different ages and stages of resorption. It can be
found throughout the retina to the ora serrata.
┬ĘVitreous haemorrhage is frequent.
┬ĘRetinal haemorrhage may be
unilateral or asymmetric in terms of number and distribution, however, some
victims have none at all (15ŌĆō25%). In severe life threatening trauma (motor
cycle, great height) retinal haemorrhage is found in less then 3%. Retinal
haemorrhages in newborns are seen in vacuum-assisted deliveries in up to 75%
and in spontaneous vaginal deliveries in up to 33%. They resolve by 2wks after
birth, at the latest by 6wks, in the great majority. A consultant
ophthalmologist with expertise in the assessment of the eyes in children
suspected to have NAHI should examine the child.
Fractures
┬ĘSince skull fractures may be
missed by bony windows on CT, a plain skull film should be obtained.
┬ĘSkull fractures do not heal by
callus formation and so dating of an injury is especially difficult. If the
edges are round and smooth it is likely to be more than 2wks old.
┬ĘIf the skull fracture is depressed
or has branching, crossing, or stellate fracture lines, it is highly suggestive
for NAI, whereas accidental fractures typically are linear, parietal, and over
the vertex.
┬ĘThe typical non-skull fracture of
child abuse is the metaphyseal fracture caused by twisting the limb. It can
also occur from birth injury (e.g. breech extraction). The ŌĆśbucket handleŌĆÖ and
ŌĆścornerŌĆÖ type metaphyseal fractures are very suggestive for NAI. It is very
important to target X-ray imaging on the metaphyses, as wider imaging can miss
fractures.
Brain imaging
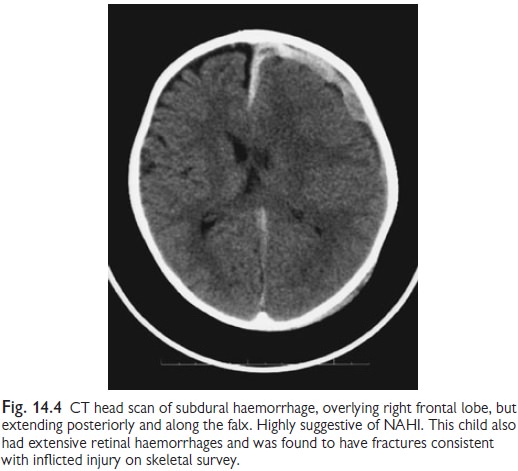
┬ĘThe initial investigation is
likely to be CT, but MRI will also be necessary in most cases (Fig. 14.4).
┬ĘMRI is more sensitive in
identifying SDHŌĆÖs of different signal characteristics, posterior and middle
cranial fossa bleeds, and parenchymal changes in the brain.
CT scans may miss small subdural
bleeds. Blood along the tentorium, interhemispheric haemorrhages, and SDHs in
multiple sites or of different densities were almost exclusively seen in NAI.
While acute haemorrhage may be isointense with brain on T1-weighted images,
acute or subacute blood is more likely to be moderately hyperintense on these
sequences.
┬Ę T2-weighted sequences may also
show high intensity, although this may be difficult to separate from the
adjacent signal in CSF. The FLAIR sequence suppresses the signal from normal
CSF, allowing the high signal haemorrhage to be visualized.
┬Ę In the early acute stage when the
blood clot is solid it may not be impressive. However, as it breaks down by
fibrinolysis and water is drawn into the haematoma a marked effusion may become
visible. Due to the dynamic changes of pathology sequential brain imaging is
recommended in order to capture the evolution of different lesions. Within
2ŌĆō4wks contusions and tears are at their most prominent.
┬Ę Encephalomalacia may be apparent
and even early atrophy. By 2ŌĆō3mths atrophy is well established. Areas of
contusion and hypoxia-ischaemia have evolved into cysts, and SDH should be
clearing.
Coagulation and haematological disorders
┬Ę For the exclusion of
thrombocytopenia, anaemia and malignancy: do platelet count, FBC and blood
film. Renal and liver function tests rule out these acquired coagulation
defects.
┬Ę The ŌĆścoagulation screenŌĆÖ comprises
of PT, APPT, Thrombin time, Fibrinogen and ŌĆśMixing studiesŌĆÖ (50:50 mix) to
exclude inhibitor.
Factor assays are available for
factor II, V, VII, VIII, IX, XI, and ŌĆśvon Willebrand`s diseaseŌĆÖ. An ╬▒ 2
antiplasmin deficiency is diagnosed by a thromboelastogram (TEG). Platelet
function disorders, vitamin C
deficiency, Factor XIII, and
collagen disorders are extremely difficult to diagnose in a child under 2yrs
and are very rare conditions.
┬ĘTherefore, investigations for
these disorders should only be done after discussion with Paediatric
haematologist, and on good clinical grounds.
Glutaric aciduria type 1 (GA1)
The exclusion of GA1 is fraught
with difficulty. The best approach is to obtain the urine and blood, however,
to delay further investigation until other investigations are back. As an
example, if the child has multiple frac-tures, or malicious injuries these
would not have been caused by GA1. In such an instance the investigations for
organic acids, acylcarnitines, or even a skin biopsy are inappropriate.
Management
┬ĘTake full social, medical, family
history, including report from social services and police on all adults in
household/caring for child.
┬ĘCT head and MRI head/spine when
possible.
┬ĘSkeletal survey.
┬ĘClotting assessment.
┬ĘTake urine to store in case of
need to check organic acids.
┬ĘArrange ophthalmology assessment.
Unless sure that there has been
accidental trauma, arrange a full confer-ence around the child with relevant
professionals, including social services, who may invite police attendance
(their responsibility). Remind them: the investigations will take at least a
week to perform, therefore there is no ŌĆśhurryŌĆÖ to produce definitive
report/guidance for other professionals until results are available.
Related Topics