Chapter: BIOLOGY (ZOOLOGY) Standard XII second year 12th text book Assignment topics question and answer Explanation Definition
Structure of a skeletal muscle, sarcomere
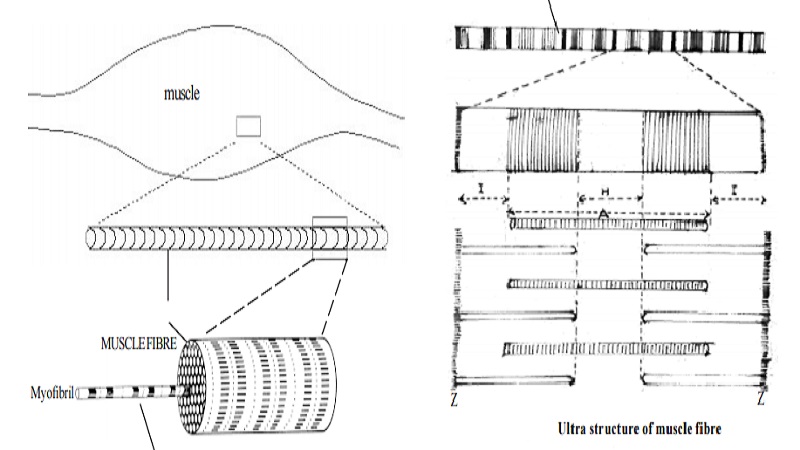
Structure of a skeletal muscle, sarcomere
Muscles
Muscles are elastic in nature. They are capable of contraction in response to stimulus from the central nervous system. The muscle cells function like biological machines that convert chemical energy into mechanical work. The mechanical work involves various movements including vital processes like contraction of the heart and blood vessels. Approximately 40% of the body is skeletal muscle and almost 10% is smooth and cardiac muscles.
Structure of a skeletal muscle:
A striated muscle is composed of many fibres arranged in bundles. The diameter of each fibre varies from 10 to 100 microns. The length of fibres ranges from 1 to 20mm. Each fibre is surrounded by a membrane, called the sarcolemma.
Each muscle fibres is made up of 4 to 20 thread-like structures called myofibrils. They are parallel to each other. The myofibrils are 1 to 3 micron in diameter. In between the myofibrils, the sarcoplasm is present. A small segment of the myofibril is called as the sarcomere.
Structure of sarcomere :
When a sarcomere is observed under a microscope, we could see alternative dense ( A band ) and light bands ( I band). The central region of the A band is often less dense and is known as the ' H Zone'. The 'I band' is bisected by a dense narrow line, the Z line. Thus each sarcomere includes repeating units between two Z lines in linear order as Z line, I band, A band, I band and next Z line.
Electron microscopic studies have shown that the striations are due to the regular arrangement of 2 types of protein filaments. 'A band' contains a set of thick filaments formed of the contractile protein myosin. It may range upto 110 Ao in diameter and 1.5 micron in length.
The second set of thin filaments ( 50 Ao diameter) overlap the long filaments in 'A band'. The second set of filaments extend partly in 'I band' and partly in 'A band'. These filaments are formed of a substance called Actin.
Myosin, actin, tropomyosin and troponin are the four major proteins which constitute the contractile machinery of muscle fibre. The energy for muscle action is provided by ATP molecules.
Related Topics