Chapter: Biochemistry: Amino Acids and Peptides
Small Peptides with Physiological Activity
Small Peptides with Physiological
Activity
What are some biological functions of small peptides?
The
simplest possible covalently bonded combination of amino acids is a dipeptide,
in which two amino acid residues are linked by a peptide bond. An example of a
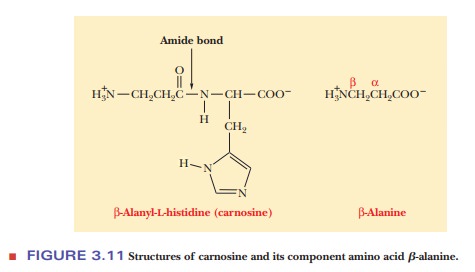
naturally occurring dipeptide is carnosine, which is found in muscle tissue.
This compound, which has the alternative name β-alanyl-L-histidine,
has an interesting structural feature. (In the systematic nomenclature of
peptides, the N-terminal amino acid
residue-the one with the free amino group-is given first; then other residues
are given as they occur in sequence. The C-terminal
amino acid residue-the one with the free carboxyl group-is given last.) The
N-terminal amino acid residue, β-alanine, is structurally different from the α-amino
acids we have seen up to now. As the name implies, the amino group is bonded to
the third or β-carbon of the alanine (Figure 3.11).

Glutathione
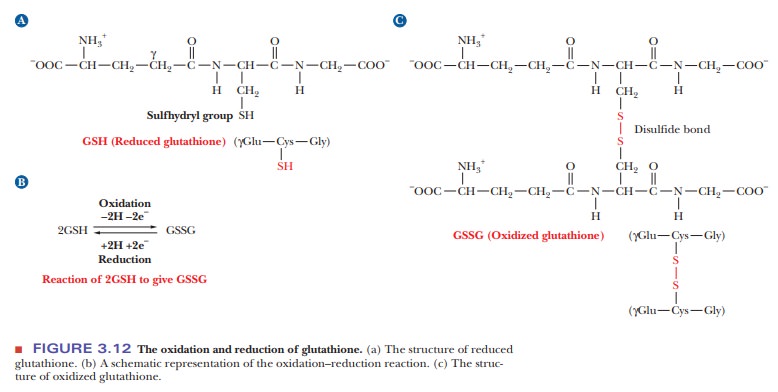
is a commonly occurring tripeptide; it has considerable physi-ological
importance because it is a scavenger for oxidizing agents. Recall that
oxidation is the loss of electrons; an oxidizing agent causes another substance
to lose electrons. (It is thought that some oxidizing agents are harmful to
organisms and play a role in the development of cancer.) In terms of its amino
acid composition and bonding order, it is γ-glutamyl-L-cyste-inylglycine
(Figure 3.12a). The letter g (gamma) is the third letter in the Greek alphabet;
in this notation, it refers to the third carbon atom in the molecule, counting
the one bonded to the amino group as the first. Once again, the N-terminal
amino acid is given first. In this case, the γ-carboxyl group (the side-chain
carboxyl group) of the glutamic acid is involved in the peptide bond; the
amino
group of the cysteine is bonded to it. The carboxyl group of the cysteine is
bonded, in turn, to the amino group of the glycine. The carboxyl group of the
glycine forms the other end of the molecule, the C-terminal end. The glutathione
molecule shown in Figure 3.12a is the reduced form. It scavenges oxidizing
agents by reacting with them. The oxidized form of glutathione is generated
from two molecules of the reduced peptide by forming a disulfide bond between
the -SH groups of the two cysteine residues (Figure 3.12b). The full structure
of oxidized glutathione is shown in Figure 3.12c.
Two
pentapeptides found in the brain are known as enkephalins, naturally occurring
analgesics (pain relievers). For molecules of this size, abbreviations for the
amino acids are more convenient than structural formulas. The same notation is
used for the amino acid sequence, with the N-terminal amino acid listed first
and the C-terminal listed last. The two peptides in question, leucine enkephalin
and methionine enkephalin, differ only in their C-terminal amino acids.
Tyr-Gly-Gly-Phe-Leu
(three-letter abbreviations)
Y-G-G-F-L (one-letter
abbreviations)
Leucine
enkephalin
Tyr-Gly-Gly-Phe-Met
Y-G-G-F-M
Methionine
enkephalin

It is
thought that the aromatic side chains of tyrosine and phenylalanine in these
peptides play a role in their activities. It is also thought that there are
similarities between the three-dimensional structures of opiates, such as
mor-phine, and those of the enkephalins. As a result of these structural
similarities, opiates bind to the receptors in the brain intended for the
enkephalins and thus produce their physiological activities.
Some
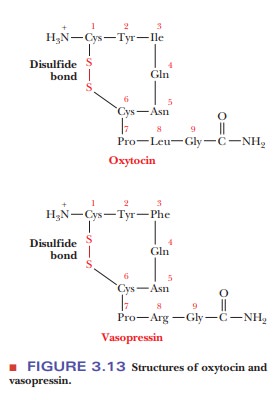
important peptides have cyclic structures. Two well-known examples with many
structural features in common are oxytocin and vasopressin (Figure 3.13). In
each, there is an -S-S- bond similar to that in the oxidized form of
glutathione. The disulfide bond is responsible for the cyclic structure. Each
of these peptides contains nine amino acid residues, each has an amide group
(rather than a free carboxyl group) at the C-terminal end, and each has a
disul-fide link between cysteine residues at positions 1 and 6. The difference
between these two peptides is that oxytocin has an isoleucine residue at position
3 and a leucine residue at position 8, and vasopressin has a phenylalanine
residue at position 3 and an arginine residue at position 8. Both of these
peptides have considerable physiological importance as hormones (see the
following Biochemical Connections box).
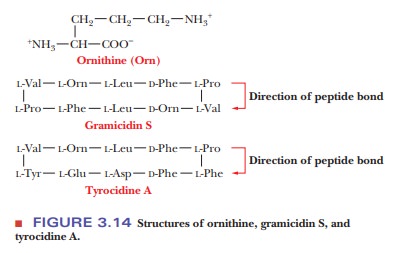
In some other peptides, the cyclic structure is formed by the peptide bonds themselves. Two cyclic decapeptides (peptides containing 10 amino acid resi-dues) produced by the bacterium Bacillus brevis are interesting examples. Both of these peptides, gramicidin S and tyrocidine A, are antibiotics, and both contain D-amino acids as well as the more usual L-amino acids (Figure 3.14). In addition, both contain the amino acid ornithine (Orn), which does not occur in proteins, but which does play a role as a metabolic intermediate in several common pathways.
Summary
Small
peptides play many roles in organisms. Some, such as oxytocin and vasopressin,
are important hormones. Others, like glutathione, regulate oxidation–reduction
reactions. Still others, such as enkephalins, are natu-rally occurring
painkillers.
Related Topics