Chapter: Obstetrics and Gynecology: Contraception
Ring and Patch - Contraception
Ring and Patch
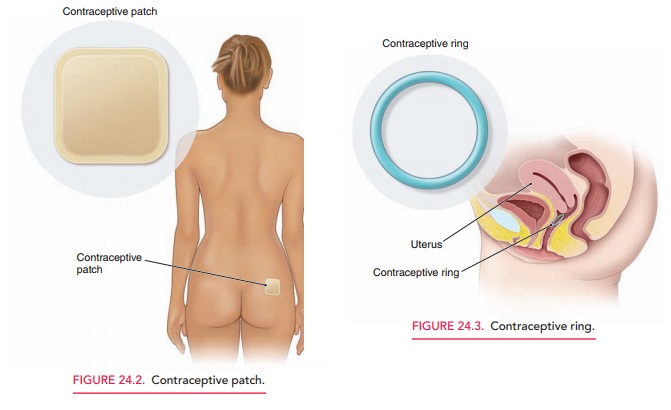
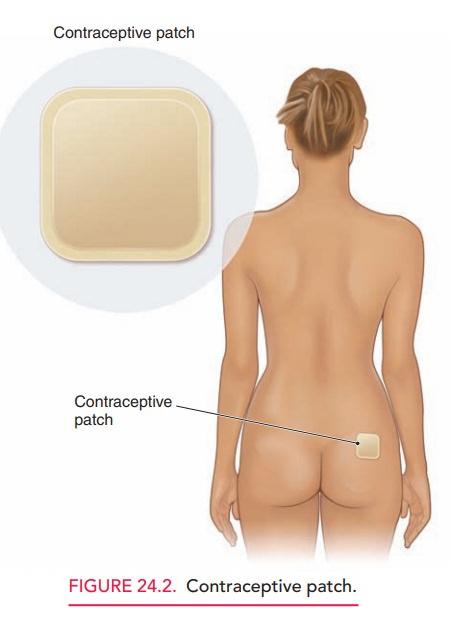
The transdermal contraceptive patch contains synthetic estrogen and
progestin and remains effective for an entire week (Fig. 24.2). The patient
should start the patch on the first day of her menstrual period and replace it
weekly for 3 weeks. The fourth week is patch-free to allow a with-drawal bleed.
Placement on clean, dry skin located on the buttocks, upper outer arm, or lower
abdomen is recom-mended. Because of its ease of application and improved
compliance, the “method failure” and “patient failure” of the patch are almost
identical. Caution should be used when prescribing the patch for women weighing
more than 90 kg (198 pounds) because of its decreased efficacy. Side effects
and contraindications are similar to the OCs. A complaint specific to the
patch, however, includes skin irritation from adhesive residue at the
application site.
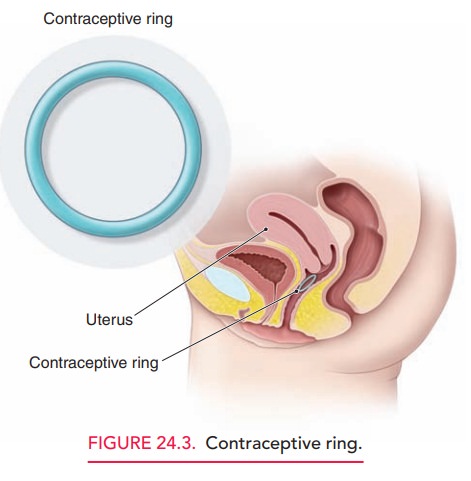
The contraceptive vaginal ring releases a sustained amount of synthetic
estrogen and progestin daily (Fig. 24.3). Comparable with oral contraceptives
in efficacy, the ring is associated with greater compliance because of its
once-a-month usage. Placed into the vagina by the patient at the beginning of
her menses, it is left in place for 3 weeks. Removal of the device results in a
withdrawal bleed. The ring can be taken out of the vagina for up to 3 hours, if
desired, without altering its efficacy. Because it is colorless and odorless,
with a 2-inch diameter, most patients and their partners are unaware of the
presence of the ring. An advantage of the ring over OCs is a decreased
incidence of break-through bleeding.
Because the hormones in the
vaginal ring and the transdermal patch do not get absorbed through the
gastro-intestinal tract, some of the medication interactions that occur with
combined oral contraceptives may not apply. However, metabolism still occurs in
the liver and, there-fore, caution must be used.
Related Topics