Chapter: Genetics and Molecular Biology: Induction, Repression, and the araBAD Operon
Regulating AraC Synthesis
Regulating AraC Synthesis
The araC gene is oriented oppositely to the araBAD genes, and its promoter, pC, is adjacent to the
regulatory sites for the BAD operon. This location raises the possibility that
the activity of pC might
be regulated by the same proteins that regulate the activity of pBAD.
Since the
measurement of AraC protein is difficult, lengthy, and imprecise, Casadaban
chose not to study regulation of the pC
promoter by measuring AraC protein itself but instead to fuse the promoter to
the
Figure
12.8 Deletion of intervening DNA fusespCto a transposedlacZ gene.
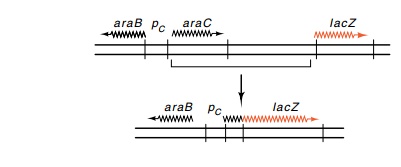
β-galactosidase gene(Fig. 12.8).
The assay of this protein is simple. Thegeneral strategy of fusing β-galactosidase to promoters has
become a widespread tool. It is used not only in the study of bacterial genes,
but also in higher cells. Its use in Drosophila,
for example, permits facile study of the spatial and temporal specificity of
gene regulation. Also, the Berk-Sharp S1 nuclease mapping method which was
discussed now permits a straightforward characterization of the pC promoter activity under a
variety of conditions. The original pC-lacZ fusion was constructed in a series
of intricate genetic operations. First the β-galactosidase gene was brought
near the araC gene and then
intervening DNA was deleted so that the structural gene of β-galactosi-dase was fused to pC. Hence the measurement of β-galactosidase became a
measurement of the activity of pC.
The first
finding was that pC is
stimulated about threefold by the presence of cyclic AMP-CRP. A more surprising
finding was that pC is
about six times more active in the absence of AraC protein than in its
presence. That is, AraC protein represses its own synthesis. A third finding
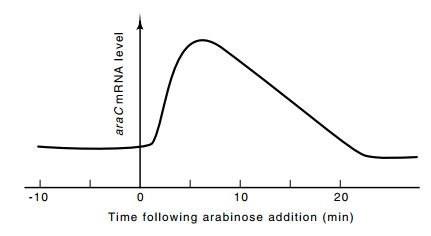
was that on the addition of arabinose to cells, the level of araC messenger increases about fourfold in several minutes and
then slowly falls back to its prior level (Fig. 12.9). Whether this transient
derepres-sion is of any physiological value is unknown. One might expect that
the resulting elevated level of AraC protein could facilitate induction of the ara operons not already saturated with
the protein.
Figure 12.9 Transient derepression ofaraCmessenger level following theaddition of arabinose to growing cells.
Related Topics