Chapter: Biochemistry: Protein Metabolism
Protein Metabolism Translation
Translation
Translation is a process by which the base
sequence of DNA transcribed to the mRNA is interpreted into amino acid sequence
of a polypeptide chain.
Translation involves the following steps:
1.
Activation
of amino acid
2.
Transfer
of activated amino acid to tRNA
3.
Initiation
of polypeptide chain
4.
Elongation
of polypeptide chain
5.
Termination
of polypeptide chain
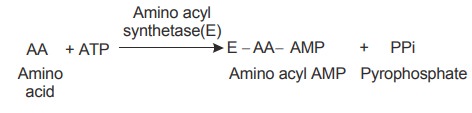
1. Activation of amino acid
Amino acids, the building blocks of proteins,
are present in the cytoplasm. They are activated before they are transported by
tRNA. The amino acids are activated by ATP with the help of the enzyme amino
acyl synthetase. Amino acyl synthetase is specific in activating each amino
acid.
The activated amino acid is called amino acyl
adenylate or amino acyl AMP.
Pyrophosphate is released.
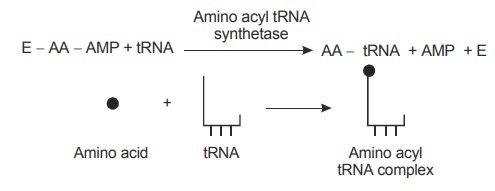
2. Transfer of activated amino acid to tRNA
The same enzyme that activates the amino acid
catalyses its transfer to a molecule of transfer RNA at the 3' hydroxyl of the
ribose, an ester with a high potential for group transfer. In this reaction AMP
and the enzyme amino acyl synthetase are released.
3. Initiation of polypeptide chain
Protein synthesis is initiated by the selection
and transfer of the first amino acid into ribosomes. This process requires
ribosome subunits, amino acyl tRNA complex, mRNA and initiation factors (IF).
Initiation of polypeptide chain involves the following steps.
1.
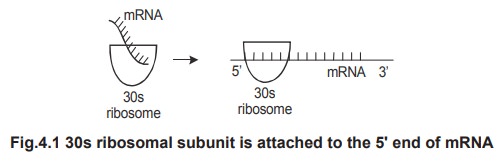
The
30s ribosomal subunit attaches to the 5' end of the mRNA to form an mRNA 30s
complex. This process requires the initiation factor IF-3 and Mg2+ ions. The
attachment is made at the first codon of the mRNA.
2.
The
first codon of mRNA will be always AUG. This codon specifies the amino acid
methionine. So the first amino acid in the synthesis of any polypeptide chain
is methionine.
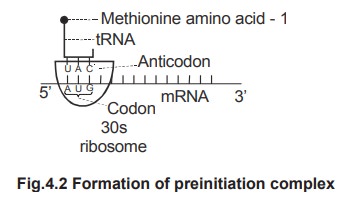
3.
The
tRNA having the anticodon UAC (complementary to AUG) transports methionine to
the 30s ribosome and attaches itself to the initiation codon on mRNA. The tRNA,
mRNA and 30s ribosome subunit form a complex called 30s - pre initiation
complex. This process requires initiation factors and GTP.
4.
30s
- pre initiation complex joins with 50s ribosomal subunit to form initiation
complex. The initiation complex is formed of 70s ribosome, mRNA and met -RNA
(methionine RNA).
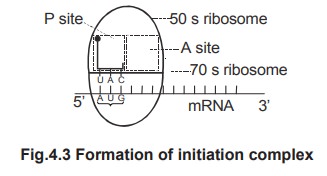
5.
The
70s ribosome has two slots for the entry of amino acyl tRNA, namely P site (peptidyl
site) and A site (amino acid site). The first tRNA i.e. met RNA is attached to
the P site of 70s ribosome.
4. Elongation of polypeptide chain
Elongation refers to sequential addition of
amino acids to methionine, as per the sequence of codon in the mRNA. It
involves the following steps:
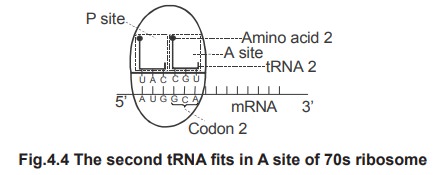
1.
The
second codon in the mRNA is recognised and as per the recognition, the amino
acyl tRNA containing the corresponding anticodon moves to the 70s ribosome and
fits into the A-site. Here the anticodon of tRNA base pairs with the second
codon of mRNA.
2.
A
peptide bond is formed between the carboxyl group (-COOH) of first amino acid
of site P and the amino group (-NH2) of second amino acid of A-site.
The peptide bond links two amino acids to form a dipeptide. The bonding is
catalysed by the enzyme peptidyl transferase which is present in 50s ribosomal
subunit.
3.
After
the formation of peptide bond, the methionine and tRNA are separated by an
enzyme called tRNA deacylase.
4.
The
dissociated tRNA is then released from P-site into the cytoplasm for further
amino acylation.
5.
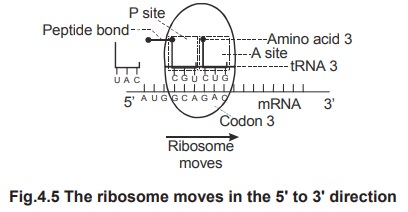
Now
the ribosome moves on the mRNA in the 5'®3' direction so that the first codon
goes out of ribosome, the second codon comes to lie in the P-site from A-site
and the third codon comes to lie in the A-site. Simultaneously, the second tRNA
is shifted from A-site to P-site. All these events, the movement of ribosome,
the release of first tRNA from P-site and shifting of second tRNA from A-site
to P-site constitute translocation. Translocation is
catalyzed by the enzyme translocase.
6.
The
third codon is recognised and the amino acyl tRNA containing the corresponding
anticodon moves to the 70s ribosome and fits into the A-site. The anticodon
base pairs with the codon. A peptide bond is formed between the third amino
acid of site-A and the second amino acid of the dipeptide present in the
P-site. Thus a tripeptide is formed.
7.
The
amino acids are added one by one as per the codon in the mRNA and hence the
tripeptide is converted into polypeptide chain. The polypeptide chain elongates
by the addition of more and more amino acids.
8.
The
elongation of polypeptide chain is brought about by a number of protein factors
called elongation factors.
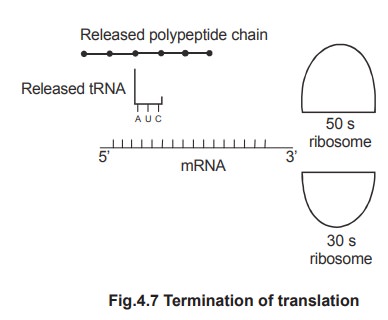
5. Termination of polypeptide chain
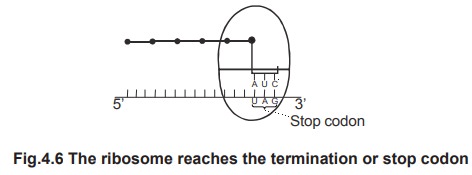
Termination is the completion of polypeptide
chain. By termination, a polypeptide chain is finished and released. The
polypeptide chain is completed, when the ribosome reaches the 3' end of mRNA.
The 3' end contains stop codons or termination
codons. They are UAG or UAA or UGA. Termination is helped by the terminating
protein factors. The terminated polypeptide chain is released from the
ribosome.
After the release of polypeptide chain, the 70s
unit dissociates into 50s and 30s sub-units. These subunits are again used in
the formation of another initiation complex.
The polypeptide chain released after translation
is inactive. It is processed to make it active. In the processing the initiating
amino acid methionine is removed.
Along with methionine a few more amino acids are
removed from the N-terminal of the polypeptide. The processing is carried out
by deformylase and amino peptidase. This processing is called as post
translational modifications.
Related Topics