Chapter: Modern Pharmacology with Clinical Applications: Adrenocortical Hormones and Drugs Affecting the Adrenal Cortex
Proposed Mechanism of Steroid Action
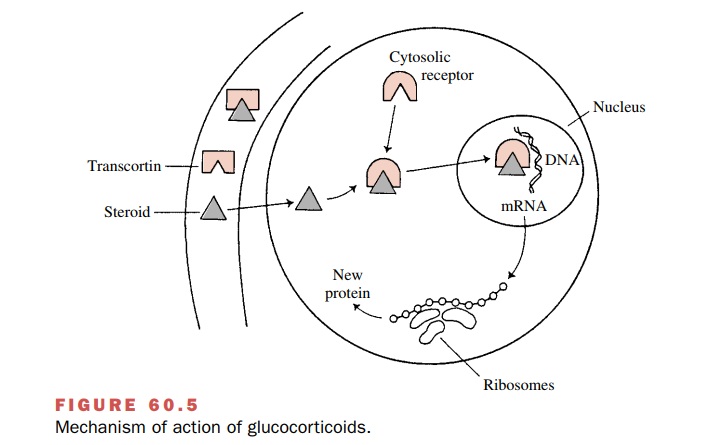
PROPOSED
MECHANISM OF STEROID ACTION
While certain properties of
glucocorticoid action are a result of direct posttranscriptional effects, most
are a consequence of effects on gene expression. Steroids transported by
transcortin enter the target cell by diffu-sion and then form a complex with
its cytosolic receptor protein. Glucocorticoids bind to cytoplasmic
glucocorti-coid receptors containing two subunits of the heat shock protein
that belong to the 90-kDa family. The heat shock protein dissociates, allowing
rapid nuclear translocation of the receptor–steroid complex. Within the
nucleus, the glucocorticoid receptor induces gene transcription by binding to
specific sequences on DNA called glucocorticoid response elements in the
pro-moter–enhancer regions of responsive genes (Fig. 60.5). In certain cases,
the glucocorticoid receptor can interact with nuclear factor- B and AP-1 to
inhibit gene expres-sion activated by these proinflammatory transcription
factors. Because their side effects are thought to be a consequence of gene
induction, glucocorticoids that can repress inflammatory genes without inducing
gene tran-scription are in development.
The pivotal role that the glucocorticoid receptor plays in hormone action is illustrated by the fact that the magnitude of induction of a regulatable gene and cellular responsiveness are directly proportional to the number of occupied receptors. A decrease in gluco-corticoid receptor number (down-regulation) produced by protein degradation may be responsible for the in-crease in steroid resistance observed clinically. Down-regulation of glucocorticoid receptors also is a potential mechanism for terminating glucocorticoid-dependent responses and for curtailing excessive cell stimulation when circulating levels of steroids are high. The effec-tiveness of glucocorticoids will also be compromised by the concomitant administration of other drugs that en-hance the clearance of glucocorticoids (ephedrine, phenytoin, rifampin). Glucocorticoids, which bind to mineralocorticoid receptors in the kidney to regulate salt balance, are inactivated by l1-β -hydroxysteroid re-ductase so that they do not elicit mineralocorticoid actions in this tissue.
However, in other tissues gluco-corticoids may exert their actions through
mineralocor-ticoid receptors. Several actions of glucocorticoids that are too
rapid to be explained by actions on transcription are mediated by effects on
membrane receptors.
Because glucocorticoids
regulate gene expression and protein synthesis, there is generally a lag of
several hours before their effects are manifest. Moreover, the duration of
various responses can endure after steroid levels fall. This may account for
the fact that side effects elicited by steroids can be minimized by
alternate-day therapy.
Metabolites of arachidonic
acid, including prosta-glandins (PG), thromboxanes, and leukotrienes, are
considered strong candidates as mediators of the in-flammatory process.
Steroids may exert a primary effect at the inflammatory site by inducing the
synthesis of a group of proteins called lipocortins. These proteins sup-press
the activation of phospholipase A2, thereby de-creasing the release
of arachidonic acid and the produc-tion of proinflammatory eicosanoids (Fig.
60.6).
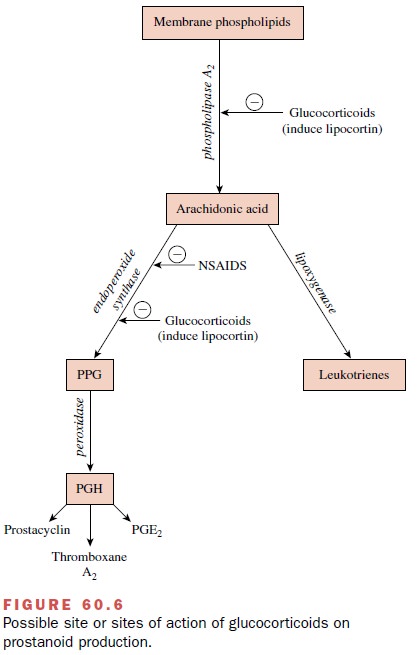
Another possible
glucocorticoid-sensitive step is the PG endoperoxide H synthase (or
cyclooxygenase) (COX) mediated conversion of arachidonate to PG en-doperoxides
(Fig. 60-6). The endoperoxides (PGG and PGH) are the precursors of PGE2,
thromboxane A2 (TBXA2), and PGI2, (prostacyclin). PG endoperoxide H
synthase has two isoforms: one is constitutively ex-pressed (PGHS-1, or COX-1),
and another is induced by growth factors, cytokines, and endotoxins (PGHS-2, or
COX-2). One component of the antiinflammatory ac-tion of glucocorticoids
appears to involve the suppres-sion of PGHS-2 induction in inflammatory cells
by proinflammatory stimuli. Inhibition of the production and effects of inflammatory
cytokines (IL-6 and IL- ) by the transcription factors nuclear factor- B and
AP-1 may also contribute to the antiinflammatory effects of glucocorticoids.
Related Topics