Chapter: Microbiology and Immunology: Bacteriology: Bacillus
Properties of the Bacteria Bacillus anthracis
Properties of the Bacteria
◗ Morphology
B. anthracis shows following morphological features:
· B. anthracis is a Gram-positive spore-forming bacillus.
· It is a very large bacillus measuring 1–1.2 mm in width and 3–5 mm in length.
· In smears from infected tissues, the bacteria are found as single, in pairs, and in short chains, the entire chain being surrounded by a capsule.
· In culture, B. anthracis grows as long chains and may appear similar to streptobacilli. In these chains, the bacilli are arranged end-to-end and the ends of the bacilli are truncated, not rounded, or often concave and somewhat swollen. This gives the chain of bacilli a “bamboo-stick” appearance (Fig. 28-1).
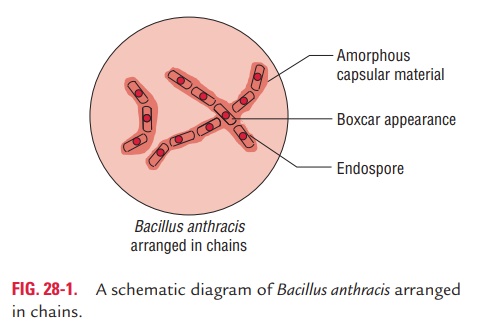
· It is capsulated. The bacterium forms the capsule only when grown on nutrient agar containing 0.7% sodium bicarbon-ate in the presence of 5–20% carbon dioxide. The capsule
It protects the bacteria against leukocytic phagocytosis and lysis. It is usually formed in the tissues but are absent when grown in ordinary conditions of culture. It is easily visual-ized using a methylene blue or India ink stain. Production of the capsule is mediated by a 60-megadalton plasmid, pX02. Therefore, the transfer of this plasmid to noncapsu-lated B. anthracis by transduction makes the recipient strain capsulated.
· It is nonmotile and nonacid fast.
Anthrax spores: Gram staining ofB. anthracisshows thecharacteristic squared ends with spores as unstained spaces. Anthrax spores are oval, central in position, and are refractile. They are of the same width as the bacillary body and so they do not cause bulging of vegetative cell. The anthrax spore consists of (a) central protoplast, (b) cortex, and (c) spore coat.
· The central protoplast or germ cell of the spore carries the constituents of the future vegetative cell. It also consists of dipicolinic acid, which is responsible for the heat resistance property of the spore.
· The cortex that surrounds the protoplast consists largely of peptidoglycan (murein), which is responsible for protecting the spore from radiation and heat. The cortical membrane, or protoplast wall, becomes the cell wall of the new vegeta-tive cell when the spore germinates.
· The spore coats, which constitute up to 50% of the volume of the spore, protect it from chemicals, enzymes, etc.
The spores are highly refractile and are resistant to staining, heat, cold, radiation, desiccation, and disinfectants. Spores are formed in culture or in soil under unfavorable conditions for growth. Anthrax spores need oxygen for sporulation. These germinate when exposed to a nutrient-rich condition, such as the tissues or blood of an animal or human host. Rainfall stimulates anthrax spore germination, while flies and vultures spread the spores. Spore formation is facilitated by:
1. nitrogen and organic soil content,
2. environmental pH greater than 6,
3. the ambient temperature greater than 15°C,
4. 2% sodium chloride, and
5. the presence of distilled water.
Spore formation is inhibited by calcium chloride. Spores are never found in host tissues unless the infected body fluids are exposed to ambient air.
◗ Culture
B. anthracis is an aerobe and facultative anaerobe. The bacteriagrow at a temperature range of 12–45°C, optimum tempera-ture being 37°C. They grow on a wide range of media including ordinary nutrient media and several selective media.
1. Nutrient agar: On nutrient agar after 24 hours of incubation,B. anthracis produces grayish and granular colonies measuring2–3 mm in diameter. Under low-power microscopy, the edges of the colony appear as long, interlacing chains of bacilli, resembling locks of matted hair, which gives them a “medusahead” appearance with an uneven surface and wavy margin.
2.Blood agar: On horse or sheep blood agar,B. anthraciscolonies are gray or white, typically nonhemolytic, with a dry, ground-glass appearance. The colonies are at least 3 mm in diameter and sometimes have tails.
3.Solid medium containing penicillin: B. anthracison a solidmedium containing 0.05–0.5 U of penicillin/mL produces large, spherical colonies within 3–6 hours and occurs in chains on the surface of the agar, resembling a string of pearls. This property is known as string of pearls reaction and is useful in differentiation of B. anthracis from B. cereus and other aerobic spore bearers.
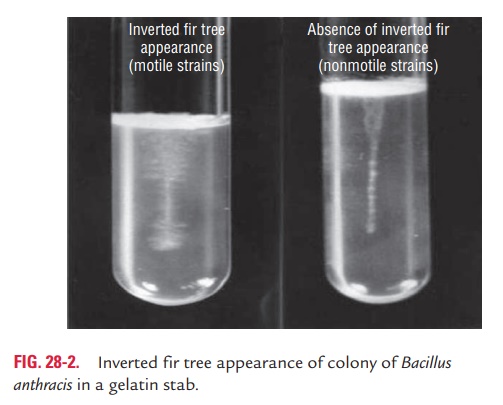
4.Gelatin medium: In a gelatin stab, there is growth downthe stab line with lateral spikes, longer near the surface, giving an ‘inverted fir tree’ appearance. The process of liquefaction is slow and late, which occurs after 7 days at 20°C and starts at the surface (Fig. 28-2).
5.Selective medium: Knisely’s Polymyxin B-lysozyme-EDTA-thallous acetate (PLET) agar medium is a selective medium used for isolation of B. anthracis from mixtures containing other spore-bearing bacilli. The medium is composed of heart infusion agar, polymyxin, lysozyme, ethylene diamine tetra ace-tic acid (EDTA), and thallous acetate.
Production of capsular material is associated with the formation of a characteristic mucoid or “smooth” (S) colony type. Capsulated bacteria on serum or bicarbonate medium produce smooth or mucoid colonies. Smooth variants that form capsule are virulent strains of B. anthracis. Rough (R) variants that lack capsule are relatively avirulent.
◗ Biochemical reactions
B. anthracis shows the following reactions:
· B. anthracis produces acid from glucose, maltose, sucrose, tre-halose and dextrin, but not from lactose, arabinose, D-xylose, or D-mannitol.
· They reduce nitrate to nitrite.
· They demonstrate a weak lecithinase reaction on egg-yolk agar, which gives a narrow zone of opalescence around the colonies.
· They are also catalase positive.
◗ Other properties
Susceptibility to physical and chemical agents: Bacillusvegeta-tive forms are susceptible to moist heat; they are killed at 60°C in 30 minutes. Bacillus spores are resistant to hostile physical and chemical conditions, ranging from desert sands and hot springs to Arctic soils and from fresh waters to marine sediments. They remain viable at temperatures, pH values, and salt concentra-tions at which few other organisms could survive. In the dry state and in certain soils, the spores may survive for 50 years or more. They survive in 5% phenol for weeks. The spores are killed by moist heat at 100°C in 60 minutes. They are also killed by 4% (w/v) formaldehyde or 4% (w/v) potassium permanganate in a few minutes.
Duckering:Destruction of the spores in animal products isachieved by duckering. Duckering is a procedure that was used to reduce anthrax spores in wool below critical infection level without destroying the animal hairs and bristles. In this pro-cedure, 2% formaldehyde is used at 30–40°C for 20 minutes for disinfection of wool, while 0.25% formaldehyde solution is used at 60°C for 6 hours for disinfection of animal hair and bristles.
Related Topics