Chapter: Medical Physiology: Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
Physiology of Deep-Sea Diving and Other Hyperbaric Conditions
When human beings descend beneath the sea, the pressure around them increases tremendously. To keep the lungs from collapsing, air must be supplied at very high pressure to keep them inflated. This exposes the blood in the lungs also to extremely high alveolar gas pressure, a condition called hyper-barism. Beyond certain limits, these high pressurescan cause tremendous alterations in body physiol- ogy and can be lethal.
Relationship of Pressure to Sea Depth. A column of seawater 33 feet deep exerts thesame pressure at its bottom as the pressure of the atmosphere above the sea. Therefore, a person 33 feet beneath the ocean surface is exposed to 2 atmos-pheres pressure, 1 atmosphere of pressure caused by the weight of the air above the water and the second atmosphere by the weight of the water itself. At 66 feet the pressure is 3 atmospheres, and so forth, in accord with the table in Figure 44–1.
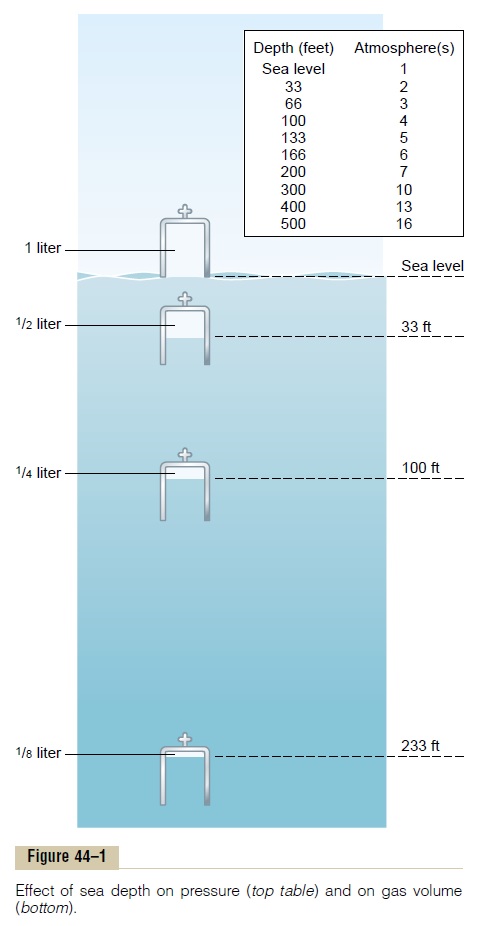
Effect of Sea Depth on the Volume of Gases-Boyle’s Law. Another important effect ofdepth is compression of gases to smaller and smaller volumes. The lower part of Figure 44–1 shows a bell jar at sea level containing 1 liter of air. At 33 feet beneath the sea, where the pressure is 2 atmospheres, the volume has been com-pressed to only one-half liter, and at 8 atmospheres (233 feet) to one-eighth liter.Thus, the volume to which a given quantity of gas is compressed is inversely proportional to the pressure. This is a principle of physics called Boyle’s law, which is extremely important in diving physiology because increased pressure can collapse the air chambers of the diver’s body, especially the lungs, and often causes serious damage.
Related Topics