Chapter: Plant Biochemistry: Phenylpropanoids comprise a multitude of plant secondary metabolites and cell wall components
Phenylpropanoids comprise a multitude of plant secondary metabolites and cell wall components
Phenylpropanoids comprise a multitude of plant secondary metabolites and cell wall components
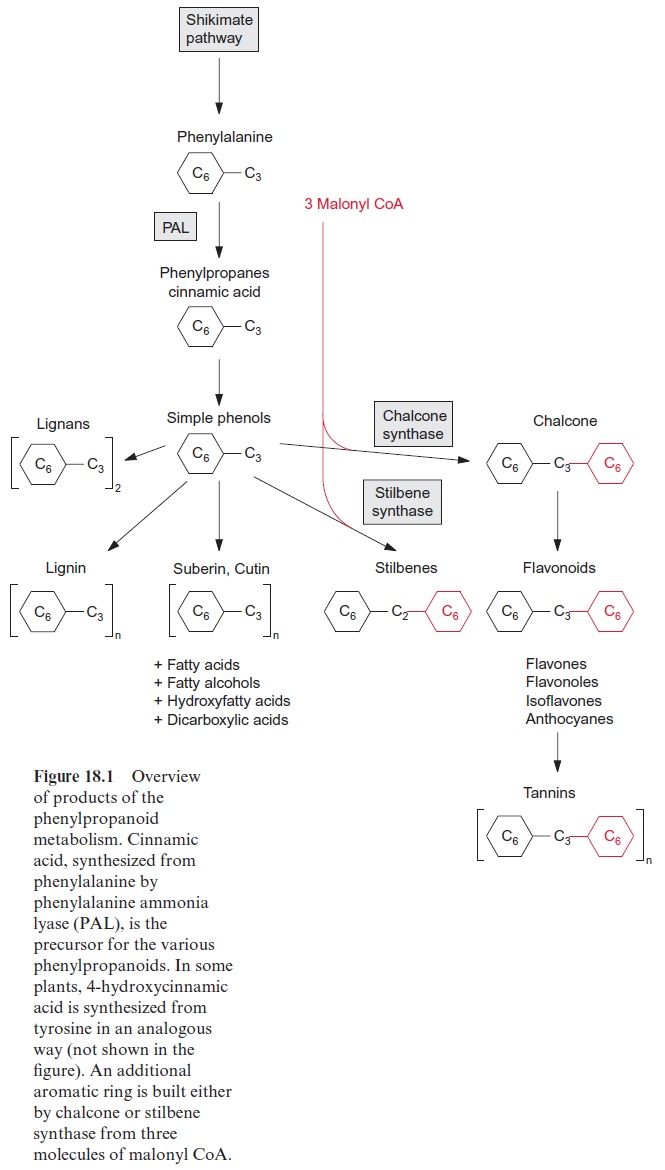
Plants contain a large variety of phenolic derivatives, which contain a phe-nyl ring and a C3 side chain and are collectively termedphenylpropanoids. As well as simple phenols, these comprise flavonoids, stilbenes, tannins, lignans and lignin (Fig. 18.1). Together with long chain carboxylic acids, phenylpropanoids are also components of suberin and cutin. These rather structurally divergent compounds have important functions as antibiotics, natural pesticides, signal substances for the establishment of symbiosis with rhizobia, attractants for pollinators, protective agents against ultraviolet (UV) light, insulating materials to make cell walls impermeable to gases and water, and structural material to assist plant stability (Table 18.1). All these substances are derived from phenylalanine, and in some plants also from tyrosine. Phenylalanine and tyrosine are synthesized by the shikimate pathway. The flavonoids, including flavones, isoflavones, and also anthocyanidins inherit the phenylpropane structure, and additionally a second aromatic ring that is built from three molecules of malonyl CoA (Fig. 18.1). This also applies to the stilbenes, but here, after the introduction of the second aromatic ring, one C atom of the phe-nylpropane is split off.

Related Topics