Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Hematopoietic Growth Factors
Pharmaceutical Issues
PHARMACEUTICAL ISSUES
Pharmaceutical issues include the status and source, storage and stability, pharmacokinetics, and pharma-codynamics of HGFs. Unless otherwise indicated, the information in this section is taken from the product package inserts.
Commercially Available Hematopoietic GrowthFactors for Clinical Use
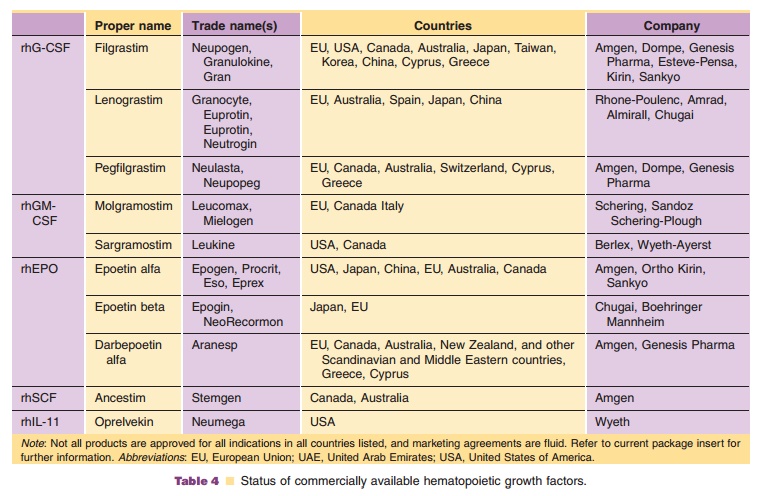
Five types of HGFs are commercially available (Table 4):
· rhG-CSF (filgrastim, lenograstim, and pegfilgrastim)
· rhGM-CSF (molgramostim and sargramostim)
· rhEPO (epoetin alfa, epoetin beta, and darbepoetin alfa)
· rhSCF (ancestim)
· rhIL-11 (oprelvekin)
Recombinant EPO, launched in 1989, was the first recombinant human HGF to be approved for marketing. Filgrastim, lenograstim, and sargramostim followed in 1991. Molgramostim was introduced in 1992. Oprelvekin was approved in 1997, ancestim in 1999, and darbepoetin alfa and pegfilgrastim in 2001. Hematopoietic growth factors may be developed by one company, manufactured by another, and distrib-uted by a third. These arrangements are dynamic, complex, and differ from country to country. Appropriate resources should be checked to deter-mine the licensing and distribution agreements in a particular country. The pharmacist, physician, or other healthcare providers are responsible for reading the current package insert for each product before prescribing, preparing, or administering any recom-binant HGF discussed in this chapter. All recombinant human growth factor preparations should be kept from freezing and should not be shaken as shaking can denature the proteins. All preparations should be visually inspected for particulate matter before administration.
Storage and Stability
Filgrastim, Lenograstim, and Pegfilgrasim
Filgrastim is available as single-use prefilled syringes and as single-use vials for subcutaneous or intrave-nous administration. Filgrastim is stable in liquid form when kept at a suitable temperature. It should be stored in the refrigerator at 2L to 8LC and should not be frozen, but accidental exposure to freezing temperatures does not adversely affect the stability. It can be left at room temperature (25LC) for up to 24 hours. Filgrastim may be diluted in 5% dextrose solution, but it should never be diluted with saline, because saline may cause the product to precipitate.
Lenograstim is available as a lyophilized pow-der for reconstitution with Sterile Water for Injection, USP. If lenograstim will be administered as an intravenous injection, it must be reconstituted in 0.9% saline or 5% dextrose solution. Reconstituted lenograstim remains stable at room temperature (25LC) for 24 hours. The lenograstim formulation contains human serum albumin.
Pegfilgrastim is supplied as prefilled syringes for subcutaneous administration. Pegfilgrastim should be stored in the refrigerator at 2L to 8LC and The reconstituted material must be used within three hours of reconstitution whether refrigerated (2–8LC) or kept at room temperature (25LC).
Related Topics