Chapter: Basic & Clinical Pharmacology : Agents That Affect Bone Mineral Homeostasis
Parathyroid Hormone - Principal Hormonal Regulators of Bone Mineral Homeostasis
PRINCIPAL HORMONAL REGULATORS OF
BONE MINERAL HOMEOSTASIS
PARATHYROID HORMONE
Parathyroid hormone
(PTH) is a single-chain peptide hormone composed of 84 amino acids. It is
produced in the parathyroid gland in a precursor form of 115 amino acids, the
remaining 31 amino terminal amino acids being cleaved off before secretion. Within
the gland is a calcium-sensitive protease capable of cleaving the intact
hormone into fragments, thereby providing one mechanism by which calcium limits
the production of PTH. A second mechanism involves the calcium-sensing receptor
which, when stimulated by calcium, reduces PTH production and secre-tion. The
parathyroid gland also contains the vitamin D receptor and the enzyme, CYP27B1,
that produces the active metabolite of vitamin D, 1,25-dihydroxyvitamin D
(1,25(OH)2D) thus enabling circulating or endogenously produced 1,25(OH)2D to
suppress PTH production. Biologic activity resides in the amino terminal region
such that synthetic PTH 1-34 (available as teri-paratide) is fully active. Loss
of the first two amino terminal amino acids eliminates most biologic activity.
The metabolic
clearance of intact PTH is rapid, with a half-time of disappearance measured in
minutes. Most of the clearance occurs in the liver and kidney. The inactive
carboxyl terminal frag-ments produced by metabolism of the intact hormone have
a much lower clearance, especially in renal failure. In the past, this
accounted for the very high PTH values observed in patients with renal failure
when the hormone was measured by radioimmunoas-says directed against the
carboxyl terminal region. Currently, most PTH assays differentiate between
intact PTH 1-34 and large inac-tive fragments, so that it is possible to more
accurately evaluate biologically active PTH status in patients with renal
failure.
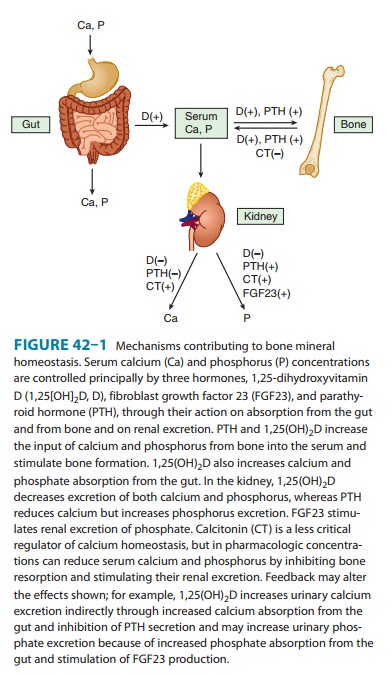
PTH
regulates calcium and phosphate flux across cellular membranes in bone and
kidney, resulting in increased serum calcium and decreased serum phosphate
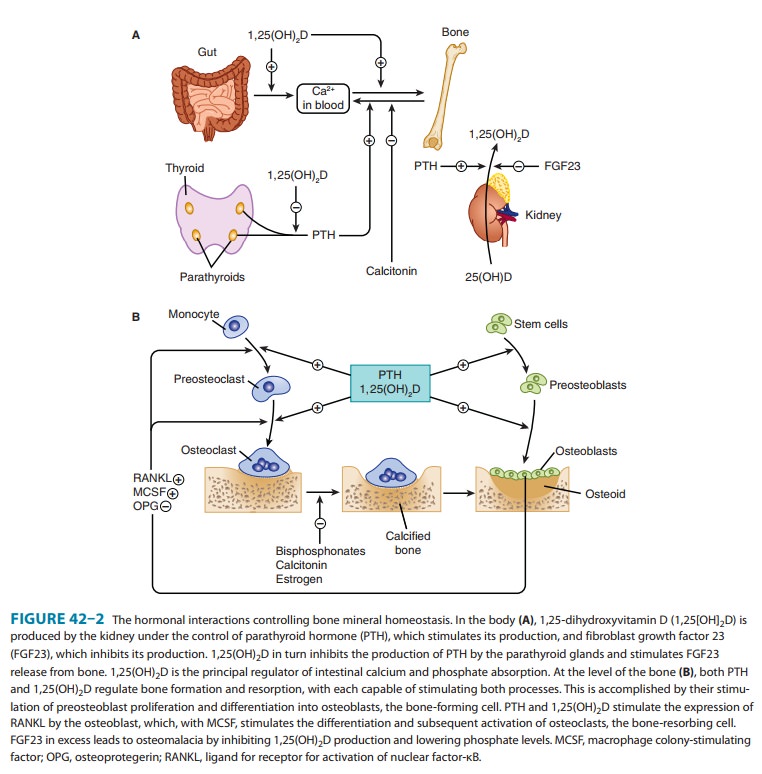
(Figure 42–1). In bone, PTH increases the activity and number of osteoclasts,
the cells
However, this stimulation of osteoclasts is not a
direct effect. Rather, PTH acts on the osteoblast (the bone-forming cell) to
induce membrane-bound and secreted soluble forms of a protein called RANKligand (RANKL). RANKL acts on
osteoclasts and osteoclast pre-cursors to increase both the numbers and
activity of osteoclasts. This action increases bone remodeling, a specific
sequence of cel-lular events initiated by osteoclastic bone resorption and
followed by osteoblastic bone formation. An antibody that inhibits the action
of RANKL has been developed (denosumab)
for the treat-ment of excess bone resorption in patients with osteoporosis and
certain cancers. Although both bone resorption and boneformation are enhanced
by PTH, the net effect of excess endoge-nous PTH is to increase bone
resorption. However, administra-tion of exogenous PTH in low and intermittent
doses increases bone formation without first stimulating bone resorption. This
net anabolic action may be indirect, involving other growth fac-tors such as
insulin-like growth factor 1 (IGF-1). It has led to the approval of recombinant
PTH 1-34 (teriparatide) for the
treat-ment of osteoporosis. In the kidney, PTH increases tubular reab-sorption
of calcium and magnesium but reduces reabsorption of phosphate, amino acids,
bicarbonate, sodium, chloride, and sul-fate. Another important action of PTH on
the kidney is stimula-tion of 1,25-dihydroxyvitamin D (1,25[OH]2D) production.
Related Topics