Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Homeostasis
Osmoregulation in different types of fishes
Osmoregulation in different types of fishes
Agnathans
Hagfishes (Myxinidae; Myxiniforms), areosmoconformers, similar to many marine invertebrates. Their overall internal osmotic concentration is about the same as that of sea water (Table 7.2). Because they live in fairly stable osmotic conditions near the bottom in relatively deep water, they do not have to contend with internal osmotic instability. Although the overall internal osmotic concentration of hagfishes is the same as the ocean, there are differences in the concentrations of some individual ions. There is no difference, however, in the concentration of the two major ions, sodium and chloride, giving hagfishes the highest concentrations of these physiologically important ions among the vertebrates. Lampreys (Petromyzontidae),the other group of extant agnathans, areosmoregulators and appear to utilize osmoregulatory strategies very similar to those of teleost’s (Evans 1993).
Elasmobranchs
To prevent osmotic stress in the hyperosmotic marine environment, marine elasmobranchs convert
Table 7.2
Plasma ionic concentrations (in milliosmoles per liter) of sea water, fresh water, and various fishes. From Evans (1993).
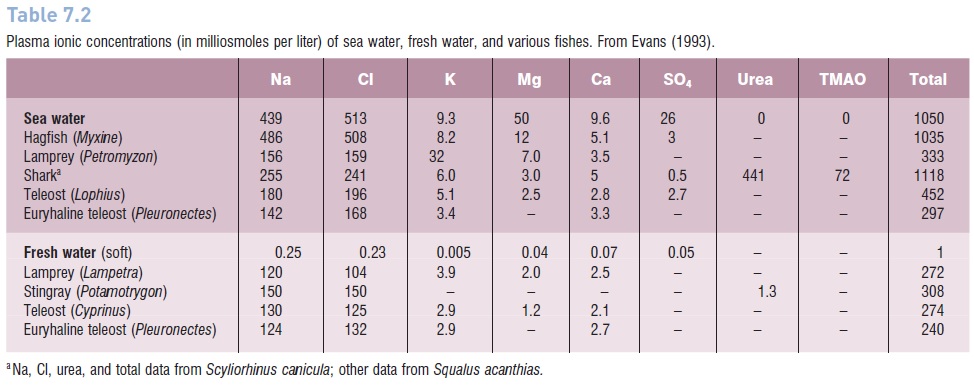
This, in addition to trimethylamineoxide (TMAO), which helps to stabilize proteins against the denaturing effect of urea, gives elasmobranch blood an osmotic concentration slightly higher than that of sea water (Table 7.1). Elasmobranch gills are not readily permeable to urea, and this is probably enhanced by the cells transporting urea back into the blood and thereby reducing the gradient between the cell and the surrounding water (Marshall & Grosell 2006). As a result of this urea retention, elasmobranchs are hyperosmotic to sea water and gain water by diffusion across their gills.
Elasmobranch gills have mitochondria-rich cells, which may help with acid–base balance but apparently play nosignificant role in sodium or chloride balance. Instead, marine elasmobranchs rid themselves of excess sodium and chloride by active secretion via the rectal gland, which lies just anterior to the cloaca. Secretory tubules of the gland are lined with salt-secreting cells that are similar structurally and biochemically to the mitochondria-rich cells of teleost gills. The rectal gland produces a solution that has about twice the NaCl concentration as the fish’s extracellular fluids (Marshall & Grosell 2006), and this solution drains into ducts leading to the lower intestine and is eliminated with other wastes.
Over 40 species of elasmobranchs, representing four families, are either euryhaline or exclusively freshwater species. Those that are euryhaline tend to lose urea when they spend time in fresh water, and those that live exclusively in fresh water, such as the freshwater stingrays(Potamotrygonidae), do not produce much urea at all and rely on ammonia excretion to get rid of nitrogen wastes, asteleosts do (Marshall & Grosell 2006). The rectal glands of these fishes are also smaller and may become atrophied due to lack of use.
Marine elasmobranchs have glomerular kidneys, and their glomerular filtration rate is somewhat similar to thoseof freshwater fishes because the high urea content of the marine elasmobranchs causes them to gain water from their environment. The nephron is long, convoluted, and has several specialized segments – the proximal segment, intermediate segment, distal segment, and collecting duct. Divalent ions, such as magnesium and sulfate, are actively transported from the blood into the proximal segment (as in marine teleost’s), and the close proximity of the looping segments suggests that a counter current mechanism may beat work, perhaps to recover urea and TMAO (Marshall &Grosell 2006). A facilitated transporter for urea has been identified in the latter segments of the nephron.
Sarcopterygians
The coelacanths (Coelacanthidae) are extant marine sarcopterygians that also maintain elevated levels of urea and TMAO in their blood to offset the high ionic concentration of the external environment (see Yancey 2001), as do the marine elasmobranchs. The African and South American lungfishes (Dipnoi) are freshwater sarcopterygians that can survive long periods of drought by estivating in mud burrows. During this estivation period they produce and retain high levels of urea, perhaps as a way of storing their nitrogen wastes in a form that is less toxic than ammonia and perhaps to help retain water. The phylogenetic distance between the sarcopterygians and the elasmobranchs, and the fact that both groups use urea as a nitrogenous wasteand osmolyte, indicates an example of convergent evolution in the face of similar physiological challenges.
Freshwater teleost’s
Freshwater teleost’s are hyperosmotic to their environment(see Table 7.1) and therefore tend to gain water and lose solutes by diffusion across the thin membranes of the gills and pharynx (Fig. 7.3A). Solutes also are lost in the urine. If left unchecked, the fish’s cells would swell and burst from the constant influx of water. To prevent this, freshwater fishes excrete a large volume of dilute urine and actively transport solutes back into their blood. Some of these solutes are recovered from urine as it is being formed in the kidney tubules. In addition, sodium and chloride ions are taken up from the surrounding water at the gills by specialized mitochondria-rich cells (often referred to as “ionoregulatory cells” or “chloride cells” in some literature). Although the specific biochemical mechanisms of this transport are not fully understood, it appears that sodium and chloride uptake are accomplished by different cells and that these processes may be linked with secretion of hydrogen ions and bicarbonate ions, respectively (Marshall & Grosell 2006). The basolateral membranes of these mitochondria-rich cells, which are essentially inward extensions of the extracellular environment, come very close to the apical surface of the cell which is in contact with the surrounding water (Fig.7.4A). This allows the transport mechanisms on the membranesto establish gradients of either hydrogen ions or bicarbonate ions high enough so that they diffuse from the cell to the surrounding water, and either sodium or chloride ions enter the cell as part of an ion exchange to maintain electrochemical balance (Fig. 7.4B, C).
The exchange of hydrogen ions for sodium ions, or bicarbonate ions for chloride ions, is likely enhanced by theenzyme carbonic anhydrase which accelerates the conversion of water and carbon dioxide to hydrogen and bicarbonate ions. Tresguerres et al. (2006) propose that the exchange of chloride for bicarbonate may be achieved by the close linkage of carbonic anhydrase and ion exchangers on the apical and basolateral membranes, a mechanism which they call the “freshwater chloride-uptake metabolon”. Sodium uptake is probably achieved by a protein that uses ATP to exchange incoming sodium ions for potassium ions (Marshall & Grosell 2006). Freshwater fishes also take up calcium at the gills by actively transporting calcium into the blood at the basolateral membrane, thereby decreasing the intracellular calcium concentration and encouraging

Figure 7.3
Maintaining osmotic balance in fresh versus seawater. (A) Freshwater bony fishes must produce a large volume of dilute urine to offset the passive uptake of water across their gills. They also must actively transport ions into the blood at the gills to compensate for the loss of these ions to the dilute freshwater environment. (B) Marine bony fishes passively lose water to their environment and gainsalts by diffusion across their gills. They must, therefore, take in water through their food and by drinking sea water. Monovalent ions are actively transported out of the blood at the gills. Magnesium and sulfate ions, which are abundant in sea water, are excreted in the urine. Marine fishes conserve water by producing urine that is isosmotic to their blood.
Kidneys also play a role in osmo regulation and ion balance. In freshwater teleost’s, glomerular filtrate passes into the proximal tubule where water and solutes, including sodium, chloride, and glucose, are recovered into the blood. Additional sodium and chloride may be recovered in the distal tubules, collecting ducts, and bladder before the urine is released from the body (Marshall & Grosell 2006).
Marine teleost’s
Marine teleost’s face the opposite problem from that of freshwater teleost’s. The high salt concentration of the ocean draws water out of the fish, and ions diffuse in across the permeable membranes (see Fig. 7.3B). To counteract potential dehydration, marine teleost’s drink sea water and actively excrete excess salts. The mitochondria-rich cells of the gills actively transport chloride ions from the fish’s extracellular fluid into the cell along the extensive basolateral membrane. This increases the chloride concentration in the cell and results in chloride diffusing out of the cell at its apical surface and into the surrounding sea water. The buildup of these negatively charged chloride ions at the outside of the apical surface attracts positively charged sodium ions, which apparently pass through the gill epithelium between the mitochondria-rich cells and the adjacent accessory cells (see Fig. 7.4D). Larger multivalent ions, especially magnesium and sulfate, which are abundant in sea water, are not readily absorbed in the gut and therefore are excreted (Marshall & Grosell 2006).
Most marine teleost’s have glomerular kidneys, so urine forms initially by glomerular filtration. Some polar fishes,however, lack glomeruli and rely exclusively on active transport of solutes from the blood into the nephron to form urine. This means of urine formation prevents the loss of important molecules, such as biological antifreezes (see above, on coping with extreme temperatures). Whether
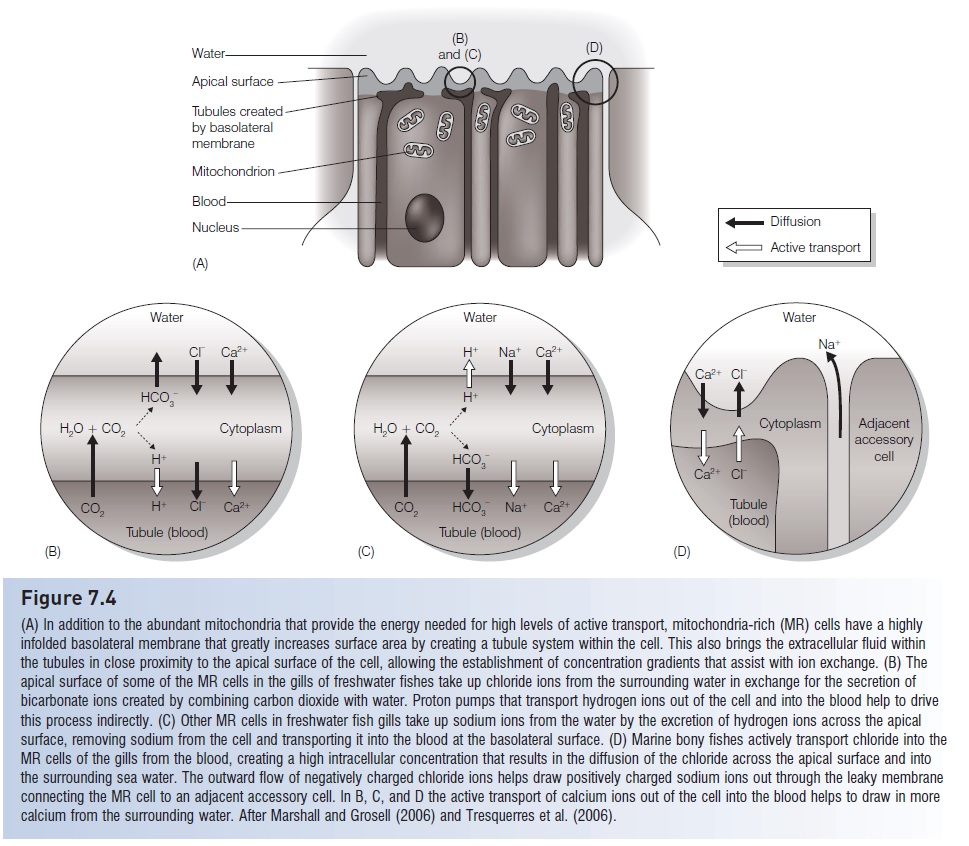
Figure 7.4
(A) In addition to the abundant mitochondria that provide the energy needed for high levels of active transport, mitochondria-rich (MR) cells have a highlyinfolded basolateral membrane that greatly increases surface area by creating a tubule system within the cell. This also brings the extracellular fluid within the tubules in close proximity to the apical surface of the cell, allowing the establishment of concentration gradients that assist with ion exchange. (B) The apical surface of some of the MR cells in the gills of freshwater fishes take up chloride ions from the surrounding water in exchange for the secretion of bicarbonate ions created by combining carbon dioxide with water. Proton pumps that transport hydrogen ions out of the cell and into the blood help to drive this process indirectly. (C) Other MR cells in freshwater fish gills take up sodium ions from the water by the excretion of hydrogen ions across the apical surface, removing sodium from the cell and transporting it into the blood at the basolateral surface. (D) Marine bony fishes actively transport chloride into theMR cells of the gills from the blood, creating a high intracellular concentration that results in the diffusion of the chloride across the apical surface and into the surrounding sea water. The outward flow of negatively charged chloride ions helps draw positively charged sodium ions out through the leaky membrane connecting the MR cell to an adjacent accessory cell. In B, C, and D the active transport of calcium ions out of the cell into the blood helps to draw in more calcium from the surrounding water. After Marshall and Grosell (2006) and Tresquerres et al. (2006).
Marine teleost also have a functionally distinct latter section of the proximal segment for the recovery of water, some sodium and chloride, and glucose. There is no distal tubule segment in marine teleost’s, so the urine passes from the proximal tubule through the collecting duct and into the bladder where additional sodium and chloride can be recovered if needed. In addition, the bladder of marine teleost’s is permeable to water, unlike that of freshwater teleost’s, providing an additional opportunity for water recovery. This is why marine teleost’s can produce urine that is isotonic with their blood, whereas freshwater teleost’s can only produce dilute urine.
Diadromous teleost’s
Teleost’s that migrate between fresh and salt water, such as salmonids, must make appropriate adjustments in the mitochondria-rich cells of the gill epithelium to physiologically adapt to the dramatic change in osmo regulatory environment. For example, as Arctic Char migrate from the ocean into rivers, the membrane proteins of the gill epithelial cells responsible for sodium–potassium exchange increase in abundance, and so do the plasma sodium concentration and blood osmolality (Bystriansky et al. 2007). This apparently is the result of increased activity of the genetic and molecular mechanisms responsible for creating these proteins, and also provides evidence that this sodium–potassium exchange plays an important role in sodium uptake in freshwater fishes. Bystriansky et al. (2007) also note that there are apparently two forms of this sodium–potassium exchange protein, one that excretes excess sodium from saltwater fishes and another that assists with sodium uptake in freshwater fishes.
Related Topics