Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: Homeostasis
Coordination and control of regulation - Fishes
Coordination and control of regulation
The nervous and endocrine systems maintain communication among the various tissues in the body and regulate many physiological functions. Neural circuitry and the speed of action potentials make the nervous system comparatively direct and fast-acting, whereas the endocrine system is better suited for long-term regulation of physiological processes because its tissues release chemical signals(hormones) into the blood. These hormones travel throughout the body, but only affect those cells with the proper molecular receptors.
The nervous and endocrine systems overlap considerably– particularly in the control of various endocrine tissues by the brain. As endocrinological research on fishes and other animals advances, it has often proven difficult to distinguish separate roles for these two regulatory systems.
The endocrine system
Ongoing research has rapidly expanded knowledge of the endocrine systems of fishes, and it is not surprising that there is great diversity in the hormones and their functions among various groups of fishes. Therefore, it is not possible given the space available to provide a complete synopsis offish endocrine tissues, their hormones, and their effects. Instead, we will provide a brief summary of some of the hormones important to homeostasis, but will not address the many other physiological functions of hormones in fishes.
Many endocrine functions are ultimately controlled by the hypothalamus of the brain regulating the many functions of the pituitary which, in turn, helps regulate many other endocrine tissues in the body. The pituitary has two main functional regions. The posterior pituitary, or neurohypophysis, is continuous with the hypothalamus and consists primarily of the axons and terminals of neurons that originate in the hypothalamus. The anterior pituitary, or adenophypophysis, lies in contact with the posterior pituitary, and in the actinopterygians the tissues fuse. The hypothalamus controls the anterior pituitary by releasing hormones delivered via blood vessels in some fishes, such as chondrichthyans, or by direct innervation as seen in some actinopterygians. Some fishes also have an intermediate lobe of the anterior pituitary, and elasmobrancheshave a ventral lobe below the anterior pituitary (Takei &Loretz 2006).
The posterior pituitary is primarily the storage and release site of chemical messengers of the hypothalamus. Neuroendocrine cells (neurons that function as endocrine cells) begin in the hypothalamus and extend into the neurohypophysis where they release their chemicals, some of which are hormones that are released into blood vesselsand trigger effects elsewhere in the body. Vasopressin (also called arginine vasotocin), for example, plays an importantrole in osmoregulation (Takei & Loretz 2006). Other chemicals released by the posterior pituitary regulate the function of cells of the adjacent anterior pituitary and intermediate lobe, and are sometimes referred to as releasing factors or releasing hormones. Some of these diffuse to the intended target cells in immediately adjacent sections of the pituitary, whereas others travel the short distance to their target cells via blood vessels.
The anterior pituitary, largely under the control of the hypothalamus, manufactures and releases hormones that control many physiological functions elsewhere in the body, including many other endocrine tissues. For example, the anterior pituitary releases adrenocorticotropic hormone(ACTH), which influences the production and release of cortisol from the interrenal tissue, and thyroid-stimulating hormone(TSH), which stimulates the thyroid gland to release thyroxin, gonadotropins (which stimulate the gonads), and growth hormone (GH) which affects various tissues throughout the body (Takei & Loretz 2006).
Fishes are the only jawed vertebrates known to possess a caudal neurosecretory system. Located at the caudal end of the spinal cord, this region of neuroendocrine cells, the urophysis, is most highly developed in the ray-finned fishes and produces urotensins that help control smooth muscle contraction, osmoregulation, and the release of pituitary hormones (Takei & Loretz 2006).
The thyroid tissue of most fishes is scattered as small clusters of cells in the connective tissue of the throat region,as opposed to the rather discrete gland found in tetrapod’s.When stimulated by TSH from the anterior pituitary, these cells produce thyroxin, which plays an important role ingrowth, development, and metabolism in many fishes. Thyroxin is quite important in development, including the sometimes extreme morphological and physiological changes associated with metamorphosis – such as the transformation of flounder from larvae with an eye on each side of the head to flatfish with both eyes on one side of the head. It also initiates seaward migratory behavior and the accompanying osmoregulatory adaptations of juvenile salmonids during their seaward spawning migration (Takei &Loretz 2006; Complex transitions: smoltification in salmon, metamorphosis in flatfish).
Maintaining proper calcium balance, including regulating calcium uptake at the gills, involves several hormones,including stanniocalcin from the corpuscles of Stannisembedded in the kidney, calcitonin produced by the ultimo branchialbodies in the back of the pharynx, and prolactinand somatolactin from the anterior pituitary (Takei& Loretz 2006).
The interrenal tissues of fishes are homologous with the distinct adrenal glands of the tetrapod’s, but are somewhatscattered in their location. The interrenal consists of two different types of cells, each of which produces different hormones. The chromaffin cells are located in the wall of the posterior cardinal vein in the pronephros of agnathans, along the dorsal side of the kidney in elasmobranchs, and in the anterior, or head, kidney of teleost’s. Chromaffin cells produce and release the catecholamine’s epinephrine (adrenaline) and norepinephrine (noradrenaline) (Takei & Loretz2006). The catecholamine’s maintain or enhance the delivery of oxygen to body tissues by increasing gill ventilation rates and blood flow, and increasing oxygen transport capability by increasing the release of red blood cells from the spleen and increasing the intracellular pH of red blood cells(Hazon & Balment 1998). This increased blood flow to the gills may lead to increased ion exchange, which may explain why stressed fishes can experience significant osmoregulatoryimbalances.
The second group of interrenal cells is that of the steroidproducing cells, located primarily in the pronephric or headkidney region. These manufacture and release corticosteroids, including cortisol, which is important in energy metabolism and maintaining electrolyte and water balance(Takei & Loretz 2006). Many other hormones also are involved in osmoregulation. For example, prolactin from the anterior pituitary, along with cortisol, is important in freshwater adaptation. Seawater adaptation involves cortisol,GH from the anterior pituitary, vasopressin from the posterior pituitary, urotensins from the urophysis, atrial natriuretic peptide from the heart, and probably others(Takei & Loretz 2006).
Glucose metabolism is influenced by insulin, glucagon, andsomatostatin from cells within the pancreas. Insulinenhances the transport of glucose out of the blood, promotes glucose uptake by liver and muscle cells, and stimulates the incorporation of amino acids into tissue proteins. Glucagon and related glucagon-like proteins seem to function in opposition to insulin, promoting the breakdown of glycogen and lipids in the liver and increasing blood glucose levels. Somatostatin also helps elevate blood glucose levels by promoting metabolism of glycogen and lipids, and by inhibiting the release of insulin (Takei & Loretz 2006).
Melatonin, produced by the pineal gland (near the top of the brain) and the retina of the eye, is secreted duringthe dark phase of daily light–dark cycles and helps regulate fish responses to daily and annual cycles of daylight. Thishormone influences many physiological processes and behaviors through its role in the maintenance of circadian activity cycles (see Circadian rhythms), daily changes in temperature preference, and changes in growth and coloration associated with changes in photoperiod and temperature (Takei & Loretz 2006).
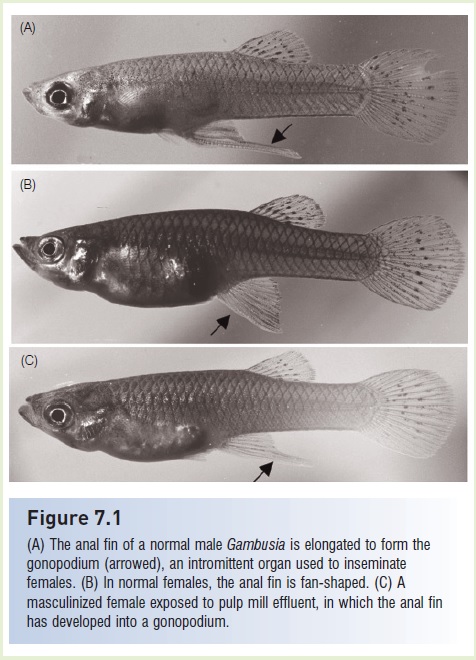
Figure 7.1
(A) The anal fin of a normal male Gambusia is elongated to form thegonopodium (arrowed), an intromit tent organ used to inseminate females. (B) In normal females, the anal fin is fan-shaped. (C) A masculinized female exposed to pulp mill effluent, in which the anal fin has developed into a gonopodium.
As briefly summarized in the preceding paragraphs, the endocrine system regulates most physiological systems associated with maintaining homeostasis. Hormones also can have a large impact on other aspects of fish biology, includingsexual development and reproductive behavior, which ultimately impact the stability of fish populations and aquatic communities. This is one reason that there has been a growing concern over human-generated endocrine disrupting compounds and their effects on development and fish population stability.
The autonomic nervous system
Involuntary physiological functions, such as control of internal organ function, are at least in part controlled by the autonomic nervous system or ANS. Neural signals from the central nervous system (brain and spinal cord)travel to ganglia of the ANS that are located either along the spinal cord or near or within the target organs. Signals then travel from these ganglia to the target tissues. The ANS is poorly developed in agnathans, better developed in elasmobranchs, and well developed in the bony fishes(Donald 2006). The ANS often works together with the endocrine system to control involuntary physiological functions such as heart rate, blood pressure, blood through the gills, and many functions of the gastrointestinal system that are important to digestion and nutrition. The ANS also controls gas bladder volume, and therefore fish buoyancy, by regulating the absorption and secretion of gases and blood flow to various parts of the gas bladder (see Buoyancy regulation, for a discussion of gas bladder function). The dispersion and aggregation of pigment inmelanophores is also partly controlled by the ANS, along with melanophore-stimulating hormone from the anterior pituitary.
Related Topics