Chapter: Genetics and Molecular Biology: Lambda Phage Genes and Regulatory Circuitry
Need for and the Realization of Hair-Trigger Induction
The Need for and the Realization of Hair-Trigger
Induction
Why does
lambda possess regulation systems as complicated as those described above? Part
of the answer is that lambda must switch between two states, lytic and
lysogenic. Even so, this does not explain why a simple repressor like that
found in the lac operon could not
suffice for lambda’s needs. In part the answer lies in the fact that lambda
must regulate its gene activities over a much greater range than lac. The basal level of the lac operon is 0.1% of the fully induced
level, and there is no apparent reason why a lower basal level would serve any
useful purpose. A similar basal level of repression of lambda early genes would
be disastrous, for lambda would then have a high rate of spontaneous induction
from the lysogenic state. Experimentally the spontaneous induction rate is low.
Typically in a doubling time, fewer than one cell out of 106
spontaneously cures of lambda or induces. This low rate means that the basal
level of expression of the lambda early genes is very low indeed.
Suppose
the basal level of expression of a repressible operon similar to the lac operon is to be generated by
increasing the concentration of repressor. To reduce the basal level to 1/1000
of normal, the repressor level would have to be raised 1000-fold. Not only
would this be wasteful of repressor, but even worse are the implications for
induction. With 10,000 molecules of repressor per cell, inducing the early
genes to greater than 50% of maximal would require inactivating approximately
of 9,998 repressor molecules per cell. Although lambda phage may like to hitch
a free ride in healthy cells, once viability of the cells is in question
because of damage to the host DNA, lambda bails out and induces. The lambda in
very few cells indeed would be capable of inducing if 99.995% of the repressor
within a cell had to be inactivated before early genes could be turned on
efficiently.
Table
14.5 Derepression of the Lambda
PromoterpRand the lac Promoter
as a Function of the Amountof Repressor Remaining Active

One of lambda’s solutions to the problem
of being either repressed or induced has been to evolve a nonlinear induction
response. At the normal levels of repressor, of about 100 dimers per cell,
lambda is fully repressed. However, if 90% of repressor has been inactivated,
the pR promoter of lambda
is 50% of fully induced. For comparison, inactiva-tion of 90% of lac repressor induces the lac operon only 3% (Table 14.5). The
highly cooperative binding of repressor to OR1
and OR2 is largely
responsible for lambda’s nonlinear response. This can be understood
quantitatively as follows. Since the high cooperativity in repressor binding
means that most often OR1
and OR2 are either
unoccupied or simultaneously occupied, it is a good approximation to assume
that two repressors bind to operator at the same time. Combining the binding
equation that defines the dissociation constant with the conservation equation
and solving yields the ratio of free operator O to total operator OT,
that is, the relative amount of derepression as a function of
repressor concentration:
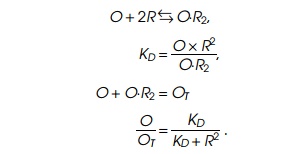
As the
concentration of free repressor falls from a value a bit above KD1/2 to a value
below KD1/2,
promoter activity, which is proportional to the fraction of operator unoccupied
by repressor, O/OT,
increases rapidly due to the R2
term. Hence very sharp changes in operator occupancy can be produced with
relatively small changes in the concentration of repressor (Fig. 14.14).
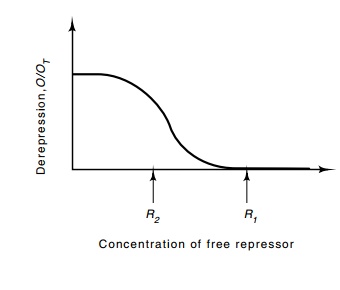
Figure 14.14 Exaggerated exam-ple of the nonlinearity in the repression of pR as a function of repressor concentration. The op-erator is fully occupied and off at concentration R1; at a repressor concentration about half of R1, the operator is unoccupied and the promoter is largely derepressed.
Lambda repressor uses another mechanism in addition to coopera-tive binding to the operators to increase the nonlinearity in its response to repressor concentration. Only dimers of the repressor polypeptide are able to bind to lambda operators under physiological conditions. The dissociation constant governing the repressor monomer-dimer reaction is such that a small reduction from the normal intracellular concentration of repressor sharply reduces the concentration of active repressor dimers. The combination of repressor monomer association-dissociation, cooperativity in repressor binding, and the need for sus-tained synthesis of N protein for lambda induction and development produces a very nonlinear induction response to changes in repressor level. At one concentration of repressor, lambda is highly stable as a lysogen, but at a lower concentration, lambda almost fully derepresses pLand pR.
Related Topics