Chapter: 10th Science : Chapter 7 : Atoms and Molecules
Molecule and Molecular Mass
MOLECULE AND MOLECULAR
MASS
Except noble gases,
atoms of most of the elements are found in the combined form with itself or
atoms of other elements. It is called as a molecule. A molecule is a
combination of two or more atoms held together by strong chemical forces
of attraction, i.e. chemical bonds.
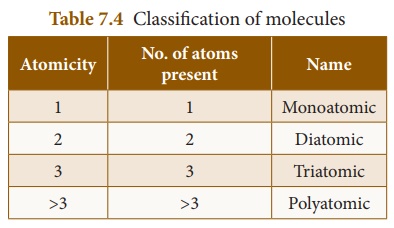
1. Classification of molecules
A molecule may contain
atoms of the same element or may contain atoms of two or more elements joined
in a fixed ratio, in accordance with the law of definite proportions. Thus, a ┬Łmolecule
may be an element or a compound. If the molecule┬Ł is made of similar
kind of ┬Łatoms, then it is called homoatomic molecule.
The molecule that
consist of atoms of different elements┬Ł is called heteroatomic molecule.
A compound is a heteroatomic molecule. The number of atoms present
in the molecule is called its ŌĆśatomicityŌĆÖ.
Let us consider oxygen. Oxygen gas exists in two allotropic forms: Oxygen (O2) and Ozone (O3). In oxygen molecule, there are two oxygen atoms. So its atomicity is two. Since both the atoms are similar, oxygen (O2) is a homodiatomic molecule. Other elements that exist as diatomic molecules are hydrogen (H2), nitrogen (N2) and halogens: fluorine (F2), chlorine (Cl2), bromine (Br2) and iodine (I2).
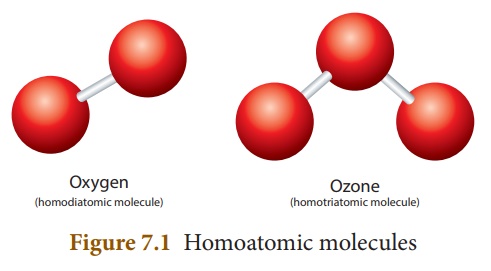
Ozone (O3)
contains three oxygen atoms and hence it is called homotriatomic molecule. If a
molecule contains more than three atoms, then it is called polyatomic
molecule.
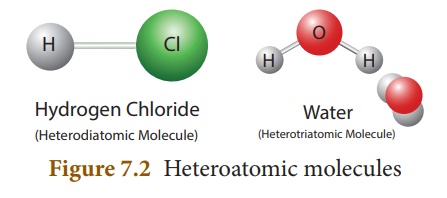
Conisder hydrogen
chloride. It consists of two atoms, but of different elements, i.e. hydrogen
and chlorine. So, its atomicity is two. It is a heterodiatomic molecule.
Similarly, the water molecule contains two hydrogen atoms and one oxygen atom.
So its atomicity is three. It is a heterotriatomic molecule.
2. Relative Molecular Mass (RMM)
As the molecules are
made of atoms, they also have their own mass. The mass of the molecule of an
element or compound is measured in the C-12 scale and hence called relative
molecular mass.
The Relative Molecular
Mass of a molecule is the ratio between the mass of one molecule of the substance
to 1/21th mass of an atom of Carbon -12.
The relative molecular
mass is obtained by adding together the relative atomic masses of all the atoms
present in a molecule.
Calculation of relative molecular mass ŌĆō Solved examples:
Example 1: Relative molecular mass
of sulphuric acid (H2SO4) is calculated as
follows: Sulphuric acid conatins 2 atoms of hydrogen, 1 atom of sulphur and 4
atoms of oxygen.
Therefore, Relative
molecular mass of sulphuric acid = (2 ├Ś mass of hydrogen) + (1 ├Ś mass of sulphur) + (4 ├Ś mass of oxygen)
= (2 ├Ś 1) + (1 ├Ś 32) +
(4 ├Ś 16)
= 98
i.e., one molecule of H2SO4
is 98 times as heavy as 1/12th of the mass of a carbon ŌĆō12.
Example 2: Relative molecular mass
of water (H2O) is calculated as follows: A water molecule is
made of 2 atoms of hydrogen and one atom of oxygen.
So, the relative
molecular mass of water
= (2 ├Ś mass of hydrogen)
+ (1 ├Ś mass of oxygen)
= (2 ├Ś 1) + (1 ├Ś 16)
= 18
i.e., one molecule of H2O is 18 times as heavy as 1/12th of the mass of a carbon ŌĆō12.
Related Topics