Chapter: Biochemical Pharmacology : Pharmacology of Eicosanoids
Lipoxygenases and related drugs
Lipoxygenases and related
drugs
The
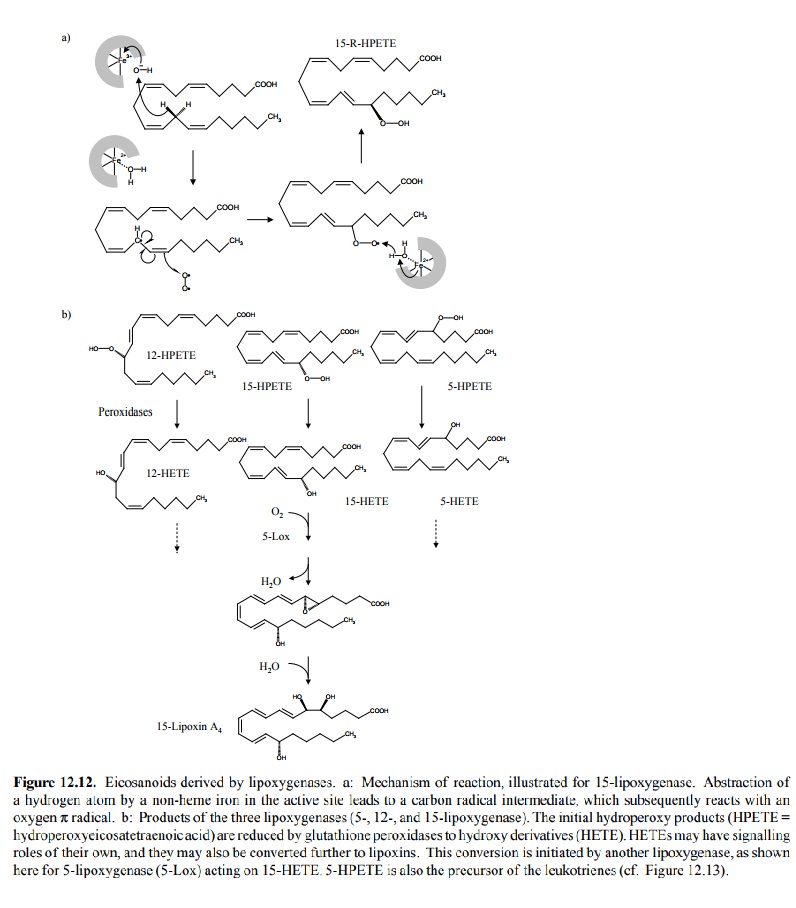
other major group of enzymes that are involved in the synthesis of eicosanoid
mediators are the lipoxygenases. There are three different such enzyme
specificities – 5-, 12-, and 15-lipoxygenase, which introduce a hydroperoxy
group at the respective carbons of arachidonic acid. Reduc-tion of these
hydroperoxides (HPETEs) leads to hydrox-yl derivatives (HETEs). These
apparently are mediators in their own rights, but they can also be further
converted to lipoxins by the successive action of different lipoxygenas-es, as
illustrated in Figure 12.12b for 15- and 5-lipoxyge-nase. Lipoxins seem to have
anti-inflammatory activity.
While the reactive groups in
the active sites of cyclooxyge-nases and lipoxygenase are quite different,
their catalyticmechanisms are actually similar in that both start out with the
abstraction of a hydrogen from the fatty acid and the subsequent combination
with an oxygen radical (cf. Fig-ures 12.6 and 12.12). Interestingly, while
Cox-1 is com-pletely inactivated by acetylsalicylic acid, acetylated Cox-2 is
still able to catalyze the above initial steps and thus function as a
lipoxygenase, giving rise to the formation of stereoisomers of physiological
HETEs and lipoxins. It has been proposed that these may contribute to the therapeutic
effect of acetylsalicylic acid.
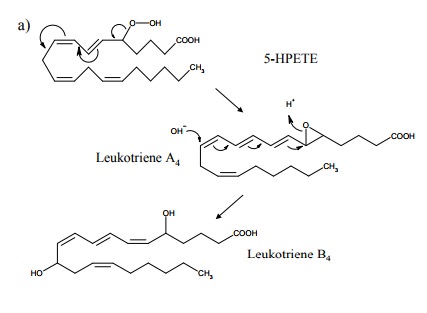
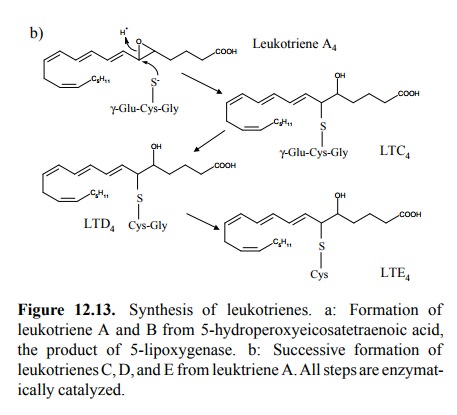
For 5-HPETE, however, there
is another important route, which leads to leukotriene A4 and from
there to either leukotriene B4 or, by reaction with glutathione and
subse-quent cleavage of peptide bonds, to leukotrienes C4, D4
and E4 (Figure 12.13). Leukotrienes (LTD4, in particular)
are potent mediators in asthma, arthritis and other chronic aller-gies and
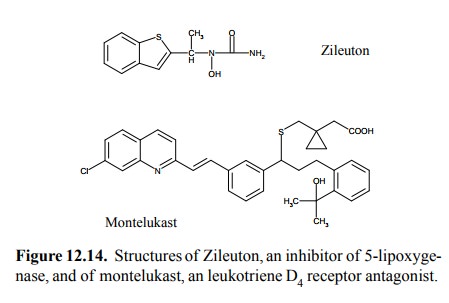
inflammatory diseases. Inhibitors of both lipoxy-genase 5 and of leukotriene
receptors are available and be-ing successfully used in clinical therapy
(Figure 12.14).
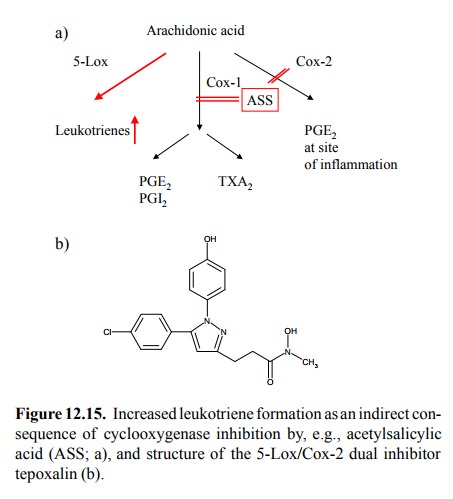
Since
lipoxygenases and cyclooxygenases share the same pool of arachidonic acid, one
might expect an undesirable increase in the synthesis of leukotrienes upon
inhibition of cyclooxygenase (Figure 12.15a). Indeed, it has long been known
that patients suffering from asthma may experi-ence clinical deterioration upon
use of acetylsalicylic acid, which is in line with a prominent role of
leukotrienes as me-diators in this disease. Selective inhibition of Cox-2
should be less freighted with this type of interaction. Moreover, there are
inhibitors (still somewhat experimental at present; Figure 12.15b) that inhibit
both 5-Lox and Cox-2 but not Cox-1 – a remarkable feat of drug development,
given that Cox-1 and Cox-2 are more closely homologous to each oth-er than to
lipoxygenase. The complementary effect – in-creased formation of prostaglandin
E and thromboxanes upon application of lipoxygenase inhibitors – has been
sub-stantiated by laboratory findings, but I am not aware of ev-idence that
this has a clinical impact.
Aside from sophisticated drug
design, there is a surprising-ly simple means of favourably influencing the
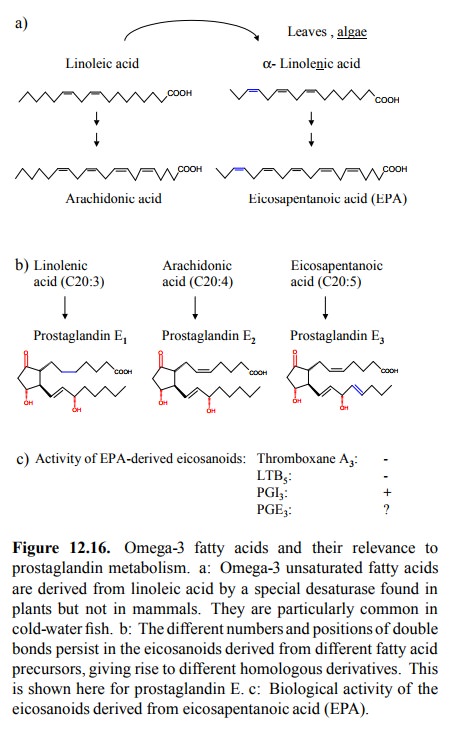
balance of Good and Evil in eicosanoid metabolism: Eating fish. As initially
mentioned, aside from arachidonic acid, other mul-tiply non-saturated fatty
acids can serve as precursors of eicosanoid mediators as well, in particular
eicosapentanoic acid (EPA), which has an additional double bond between C17 and
C18. The introduction of this `omega-3' double bond7 does not occur,
nor is it reversible, in mammalian metabolism (Figure 12.16a). It does,
however, occur in plants, where it contributes to the adjustment of membrane
fluidity in cold environments. Accordingly, we find high concentrations of EPA
in algae from cold water, and from the animals (fish) feeding on them. A high
intake of this type of food will lead to a partial replacement of arachidon-ic
acid in our own cell membranes by EPA, and therefore to an increased fraction
of EPA derivatives in our eicosanoids (Figure 12.16b). Some of the EPA derived
mediators are as active as those derived from arachidonic acid. However, it
appears that this does not apply to thromboxane (TXA3) and
leukotrienes (LTD5 and similar). Since these are the major culprits
among the eicosanoids in sustaining inflammation and triggering thrombocyte
aggregation, the result of a diet rich in fish and seaweed should be a
reduction in inflam-mation and thrombocyte aggregation, a longer life, grumpy
heirs, and impoverished physicians.
Now,
does this work? There are some intriguing statis-tics to support this: While in
western countries 40% of all deaths are due to atherosclerosis (in which both
inflamma-tion and thrombocyte aggregation are important), this num-ber stands
at 7% among Greenland eskimos and at 12% in Japan; both countries have more
fish and less meat in their diets. However, it should go without saying that we
cannot precisely gauge the contribution of EPA and related fatty acids to this
remarkable statistics.
Related Topics