Chapter: Microbiology and Immunology: Virology, Virus: Orthomyxoviruses
Laboratory Diagnosis - Influenza Viruses Infections
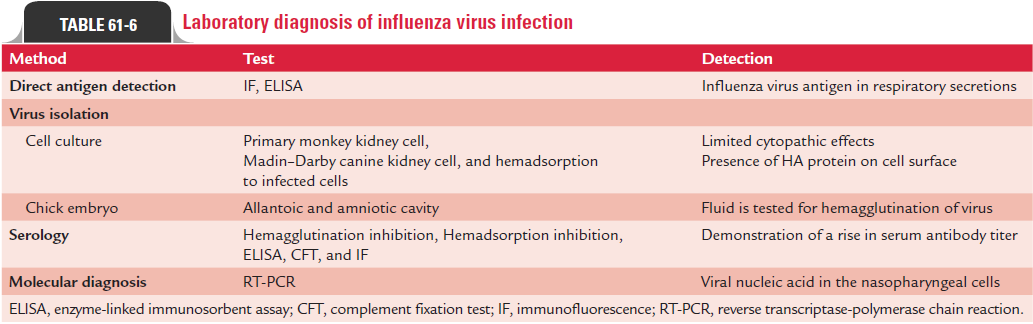
Laboratory Diagnosis
During an epidemic of influenza, the clinical diagnosis can be made, but definitive diagnosis depends on the laboratory methods.
◗ Specimens
Specimens include nasal or throat washings or sputum for viral antigen and viral RNA, throat gargles for isolation of viruses, and serum for viral antibodies.
◗ Direct antigen detection
A rapid, specific diagnosis of influenza is made by demonstrating viral antigens directly on cells obtained from the nasopharynx. Immunofluorescence (IF) or enzyme-linked immunosorbent assay using specific monoclonal antibodies are used commercially to detect viral antigen. The results of the rapid tests are useful to start treatment with the NA inhibitors within 48 hours of the onset of symptoms.
◗ Isolation of the virus
Throat gargles are the specimen of choice. The specimen is collected in saline broth or a buffered salt solution and is sent immediately to the laboratory, or if delayed is stored at 24°C. The virus is isolated from the specimen by inoculation into embryonated eggs or into certain cell cultures.
Egg inoculation: The specimen is inoculated into the amni-otic cavity of the chick embryo. After incubation at 35°C for 3 days, the amniotic fluid and allantoic fluid are harvested and tested for the presence of viral HA. This is carried out by using fowl and guinea pig red cells in parallel and incubating at room temperature and at 4°C. Usually, influenza A viruses aggluti-nate only guinea pig cells, influenza B both fowl and guinea pig red cells, and influenza C agglutinate only fowl cells at 4°C. If the test is positive, the isolate is then typed by a serological test (e.g., hemagglutination inhibition test) using specific anti-sera to types A, B, and C.
Cell culture: Influenza virus is usually isolated from respira-tory secretions by growing in tissue cultures (monkey kidney or baboon kidney cell lines). The cell cultures are incubated at 33°C in the roller drums in the presence of trypsin, but without serum.
Virus growth in tissue cultures is detected by direct demon-stration of viral antigen in infected cell cultures by IF or by testing for hemadsorption with human O, fowl, and guinea pig red cells. In a positive hemadsorption test, red cells adhere to the virus budding from infected cells. If the culture tests positive, serologic tests with specific antisera may be used to identify the virus.
◗ Serodiagnosis
Demonstration of a rise in serum antibody titer between acute-phase and convalescent-phase sera by a serological test is diag-nostic of infection. The acute-phase sera are collected within a few days of illness and the convalescent sera 7–10 days after the illness. Complement fixation tests (CFTs) with RNP antigens of influenza types A, B, and C and also the CFTs with V anti-gens are employed for demonstration of rising antibody titer in the paired sera samples.
Hemagglutination inhibition test, enzyme neutralization test, radial immunodiffusion test, and ELISA are the other tests also used for demonstration of antibodies. However, none of these techniques are useful to identify all infections. Various approaches followed for laboratory diagnosis of influenza are summarized in Table 61-6.
Related Topics