Chapter: Nutrition and Diet Therapy: Minerals
Iron (Fe) - Nutrition Trace Minerals
TRACE MINERALS
Iron (Fe)
The
principal role of iron is to deliver oxygen to body tissues. It is a component
of hemoglobin, the coloring matter of red blood cells (erythrocytes).
Hemoglobin allows red blood cells to combine with oxygen in the lungs and carry
it to body tissues.
Iron is also a
component of myoglobin, a protein compound in
muscles that provides oxygen to cells, and it is a constituent of other body
compounds involved in oxygen transport. Iron is utilized by enzymes that are
involved in the making of amino acids, hormones, and neurotransmitters.
Sources.Meat, poultry, and fish are the best sources
of iron because onlythe flesh of animals contains heme iron. Heme iron is
absorbed more than twice as efficiently as nonheme iron. Nonheme iron is found
in whole-grain cereals, enriched grain products, vegetables, fruit, eggs, meat,
fish, and poultry. The rate of absorption of nonheme iron is strongly
influenced by dietary factors and the body’s iron stores. Factors affecting the
absorption of both heme and nonheme iron are listed in Table 8-7.
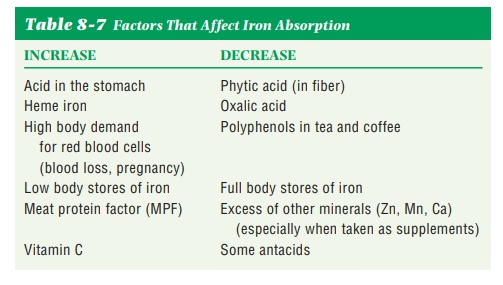
For iron to be
absorbed, it must be chemically changed from ferric to ferrous iron. This
change is accomplished by the hydrochloric acid in the stom-ach. Absorption of
nonheme iron can be enhanced by consuming a vitamin C–rich food and a nonheme
iron–rich food at the same meal. Vitamin C holds onto and keeps the iron in its
ferrous form, which facilitates absorption. Meat protein factor (MPF) is a
substance in meat, poultry, and fish that aids in the absorption of nonheme iron.
Phytic acid and oxalic
acid can bind iron and reduce the body’s absorp-tion of it. Polyphenols, such
as tannins in tea and related substances in coffee, also reduce the absorption
of iron. Antacids containing calcium and calcium supplements should be taken
several hours before or after a meal high in iron because calcium also
interferes with iron absorption.
Requirements.The NRC has determined that men lose
approximately1 mg of iron a day and that women lose 1.5 mg a day. On the
assumption that only 10% of ingested iron is absorbed, the DRI for men has been
set at 10 mg and for women from the age of 11 through the childbearing years at
15 mg. This is doubled during pregnancy and is difficult to meet by diet alone.
Consequently, an iron supplement is commonly prescribed during pregnancy. Women
should make a special effort to include iron-rich foods in their diets atall
times. The rapid growth periods of infancy and adolescence also produce a heavy
need for iron.
Deficiency or Toxicity.Iron deficiency continues to be a
problem,especially for women. Iron deficiency can be caused by
insufficient intake, malabsorption, lack of sufficient stomach acid, or
excessive blood loss, any or all of which can deplete iron stores in the body.
Decreased stores of iron pre-vent hemoglobin synthesis. The result is an
insufficient number of red blood cells to carry needed oxygen. What begins as
iron deficiency can become iron deficiency
anemia. Iron deficiency anemia takes a long time to de-velop, but it is
the most common nutrient deficiency worldwide. Symptoms include fatigue,
weakness, irritability, and shortness of breath. Clinical signs include pale
skin and spoon-shaped fingernails.
Some people suffer
from hemochromatosis. This is a
condition due to an inborn error of metabolism and causes excessive absorption
of iron. The onset of this disorder can happen at any age. Unless treated, this
condition can dam-age the liver, spleen, and heart. To control the buildup of
iron, patients with this condition must give blood on a regular basis.
Related Topics