Natural and Synthetic indicators | Acids and Bases | Chapter 14 | 8th Science - Indicators | 8th Science : Chapter 14 : Acids and Bases
Chapter: 8th Science : Chapter 14 : Acids and Bases
Indicators
Indicators
An indicator or acid– base indicator is a chemical substance which indicates the acidic or basic nature of a solution by suitable colour change. These may be natural or synthetic.
1. Natural indicators
Natural indicators are chemical substances which are obtained from the natural resources. Litmus, turmeric juice, China rose petals, red cabbage, grape juice and beetroot juice are the indicators obtained from natural resources.
Turmeric indicator
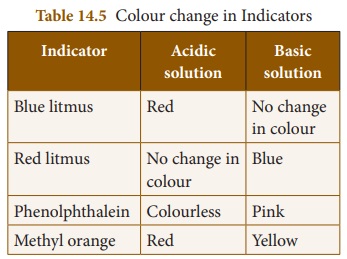
By adding small amount of water to turmeric powder a paste is prepared. This is applied on a blotting paper or filter paper and dried. These strips are used as indicators to find the nature of the solution. In acidic solution turmeric indicator paper has no change in colour. That means it remains yellow. In basic solution the colour changes from yellow to red.

Activity 4
Take a white cloth with turmeric powder stain. Wash the cloth with washing soap. Do you observe any change in the colour? Why?
Answer: Yes, the colour changes from yellow to red, because soapy solution is a base.
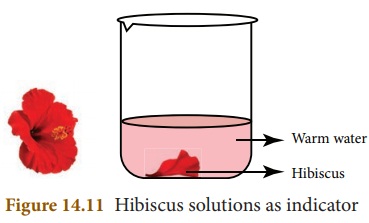
Hibiscus flower indicator
Some hibiscus flowers soaked in warm water for about 5 to 10 minutes forms a solution. This solution can be used as indicator. In acidic solution, the colour will be changed to deep pink or deep red. In basic solution, the colour will be changed into green.
Beet root juice indicator
Extracts of beet root are also used as an indicator for identifying the acidic or basic nature of a solution.
Activity 5
Take a small beet root vegetable and cut it into pieces. Boil them in hot water and filter the extract. Take two test tubes. Take sodium hydroxide solution in one test tube and vinegar or lemon juice in another test tube. Add beet root extract slowly. Observe the colour change. What do you infer?
Answer:
(i) Observation : When beetroot juice is added with sodium hydroxide solution it turns into greenish yellow shows that NaOH - is a base.
(ii) When it is added with lemon juice, the colour of beetroot juice remains same shows that lemon juice is acidic.
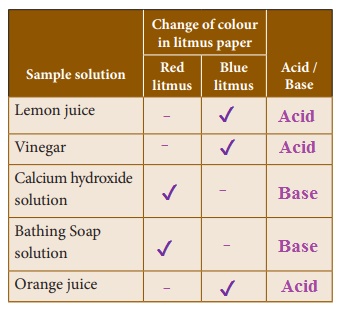
Litmus
Litmus is the most common indicators used in the laboratories. Litmus is a natural indicator which is extracted from lichens.

It is available in the form of solution or in the form of strips prepared by absorbing litmus solution on filter paper. It is either red or blue in colour. Blue litmus paper turns red in acidic solution and red litmus paper turns blue in the basic solution.
ACTIVITY - 6
Find out the nature of the solution.
2. Synthetic indicators
An indicator prepared from artificial substances is known as synthetic indicators. Phenolphthalein and methyl orange are the examples for synthetic indicators.
Phenolphthalein
Phenolphthalein is a colourless compound. Its alcoholic form is used as an indicator. It is colourless in acidic solution but turns pink in basic solution.
Methyl orange
Solid methyl orange is dissolved in hot water and its filtrate is used as an indicator. It turns red in acidic solution and yellow in basic solution.
The following table gives the colour changes of different indicators in acidic and basic medium.
Related Topics