Chapter: 11th Nutrition and Dietetics : Chapter 10 : Proteins and Lipids
Hydrogenated fats
Hydrogenated fats
Hydrogenation (or,
more accurately, "partial hydrogenation," as the process is
incomplete) is the forced chemical addition of hydrogen into omega-6
polyunsaturated oils to make them hard at room temperatures, primarily as a
cheaper and less perishable substitute for butter. The liquid fat becomes a
solid fat and the unsaturated fatty acid contents decrease as a result of
hydrogenation. Common hydrogenated fats include hydrogenated or partially
hydrogenated cottonseed, palm, soy and corn oils, but theoretically almost any
polyunsaturated oil can be hydrogenated. During the
process
of
hydrogenation,
hydrogen is added to the unsaturated linkage with nickel as
catalyst.
A major health concern
during the hydrogenation process is the production of trans fats. Trans fats
are the result of a side reaction with the catalyst of the hydrogenation
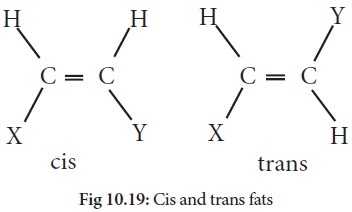
process. This is the result of an unsaturated fat which is normally found as a
cis isomer converts to a trans isomer of the unsaturated fat. Isomers are
molecules that have the same molecular formula but are bonded together
differently. A cis isomer has the hydrogens on the same side, whereas a trans
isomer has hydrogen atoms on the opposite side. Due to the added energy from
the hydrogenation process, the activation energy is reached to convert the cis
isomers of the unsaturated fat to a trans isomer of the unsaturated fat .
Although trans fats
are edible, consumption of trans fats has been shown to increase the risk of
coronary artery disease in part by raising levels of the lipoprotein LDL(often referred to as "bad
cholesterol"), lowering levels of the lipoprotein HDL (often referred to
as "good cholesterol"), increasing triglycerides in the bloodstream
and promoting systemic inflammation. Trans fat are found in margarine,
vanaspathi, baked goods such as doughnuts, pastries, cookies, deep fried foods
like fried chicken and French-fried potatoes, microwave popcorn, snack chips,
processed foods and confectionery fats.

Related Topics