Chapter: Surgical Pathology Dissection : The Cardiovascular, Respiratory System
Heart Valves: Surgical Pathology Dissection
Heart Valves
For
years, almost all valvular heart disease has been ascribed to chronic rheumatic
heart disease. As a result, excised heart valves are among the most neglected
specimens in the surgical pathol-ogy laboratory. Failure to pay appropriate
atten-tion to resected valves can be a disservice to the patient. Careful
examination of heart valves not only will help in the clinical management of
these patients but may also help in the development of improved prosthetic
heart valves.
As is
true for any specimen, clinical informa-tion is essential for the appropriate
classification of heart valve disease. The results of echocardiog-raphy and
cardiac catheterization, as well as the surgeon’s operative findings, should
all be ob-tained before you begin your examination. Al-though there is some
overlap, the examination of native, mechanical, and bioprosthetic heart valves
each presents unique challenges.
Native Heart Valves
Although
we tend to think of heart valves as consisting only of the valve leaflets,
remember the other components of the valve. The atrioven-tricular valves
(mitral and tricuspid) are com-posed of an annulus, leaflets, chordae
tendineae, and papillary muscles. The semilunar valves (the aortic and
pulmonic) are made up of three cusps, each with a sinus. These cusps meet at
three commissures. Remembering these basic components of each valve is essential,
because, although some native heart valves can be re-moved intact by the
surgeon, the majority are fragmented during removal.
Before
handling an excised valve, check to see if cultures are needed. In addition, a
photograph of the inflow and outflow aspects of a valve can help document the
cause of the disease. Similarly, a radiograph of the specimen may help
establish the extent and distribution of calcifications. Begin by documenting
whether the valve is in one piece or is fragmented. Note the dimensions of the
valve as well as the dimensions of the valve ori-fice. Next, systematically
examine each compo-nent of the valve. Start with the leaflets. Count and record
the number of leaflets. Note the edges of the leaflets, and look for any
evidence of rolling or commissural fusion. Next, examine the leaflets
themselves. Document the presence or absence of myxoid changes, fibrosis,
calcifications, thrombi, and vegetations. If the leaflets are fibrotic, are
they diffusely or focally involved? If calcifications or thrombi are present,
document their location, size, and apparent impact on valve function. If
vegetations are noted, are they friable or firm? Next, for atrioventricular
valves, examine the chordae tendineae and papillary muscles. Are the chordae normal,
or are they shortened, thick-ened, stretched, fused, or ruptured? Are the
pap-illary muscles normal, or is there evidence of recent or remote myocardial
infarction? If a por-tion of the annulus is present, examine it as well. Once
you have completed your gross descrip-tion, submit a section for histology. The
section should include the valve leaflet, the free edge of the valve, and if
present the chordae and pap-illary muscle. It may be necessary to decalcify
this section.![]()
Mechanical Heart Valves
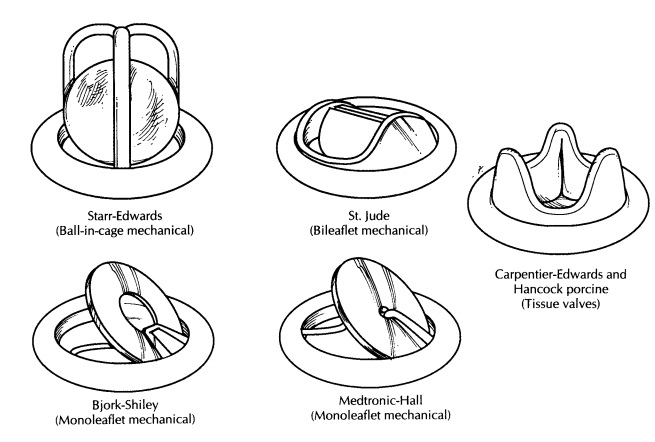
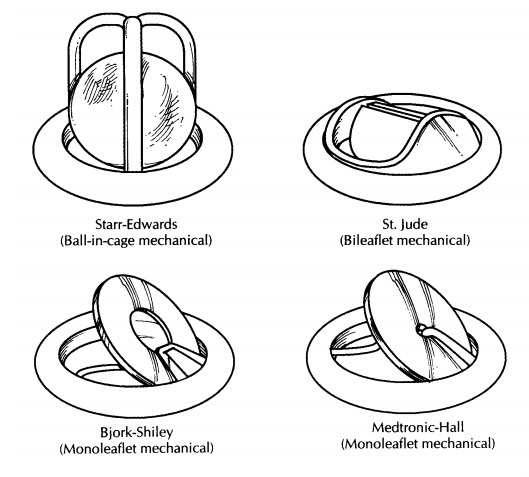
Numerous different types of mechanical heart valves have been developed over the years. The majority of these valves have three main compo-nents (1) a cloth ring, which is used by the sur-geon to sew the valve in place; (2) an occluder or poppet (ball or disk); and (3) components that limit the movement of the poppet or allow the occluder to tilt. Despite the similarities, the vari-ous types of mechanical heart valves have sev-eral important differences, and different valves are prone to different complications. Therefore, before beginning your examination of a mechani-cal heart valve, we recommend that you identify the type of valve by referring to a valve identifica-tion guide such as ‘‘Cardiac Valve Identification Atlas and Guide,’’ in Guide to Prosthetic CardiacValves.5Examples of some of the more commontypes of mechanical and bioprosthetic valves are illustrated here.
Next,
the valve can be cultured, photo-graphed, and x-rayed as indicated. Try out the
movement of the valve. Does the valve appara-tus open and close fully? Thrombi
can form at the junction of the various components of the valve, especially at
cloth-metal interfaces. These thrombi are important to identify, because they
can be a source of emboli or a nidus for the development of an infection, and
they may in-terfere with valve function. Document the loca-tion and size of any
thrombi or vegetations, and note whether they interfere with valve func-tion.
Some valves incite an intense overgrowth of fibrous tissue, and this fibrous
tissue can cause valve dysfunction and even luminal stenosis. Therefore, it is
important to note the presence or absence of fibrous tissue. Finally, carefully
look for evidence of wear and tear on the various me-chanical components of the
valve. Is there any evidence of cracking or disk wear? As is true for natural
heart valves, the effect of any pathology on valve function should be carefully
docu-mented. In most cases, it is not possible to submit any tissue for
histologic examination; but if vege-tations are present, a section should be
submitted for histology. In the United States, the U.S. Safe Medical Devices
Act of 1990 (Public Law 101-629) requires you to notify either the manufacturer
or the Food and Drug Administration if you dis-cover that a malfunctioning
prosthetic valve has contributed to the harm or death of a patient. Finally, as
is true for all mechanical devices re-moved surgically, be sure to save the
valve because it may need to be returned to the manu-facturer.
Bioprosthetic Heart Valves
A variety of bioprosthetic heart valves are used. These include (1) porcine aortic valves, (2) bovine and pericardial valves, and (3) human aortic homographs. As is true for the native and mechan-ical heart valves, the first step should be to ask yourself if the valve needs to be cultured, photo-graphed, or x-rayed. Next, carefully inspect the valve leaflets for thrombi, vegetations, calcifica-tions, and evidence of fibrous overgrowth. In par-ticular, look for evidence of tears or perforations in the valve leaflets. Again, make sure to docu-ment both the location and size of any lesions and to describe the effect of these lesions on valve function. If a sewing ring is present, be sure to examine it as wel.
Left Ventricular Assist Devices
Examination
of left ventricular assist devices re-quires special tools that may need to be
provided by the manufacturer. In general, a pump is con-nected to conduits that
contain artificial valves (usually bioprostheses), which in turn connect to the
left ventricular apex and the aorta. These devices can become infected, and it
is the respon-sibility of the pathologist to make every attempt to document any
sites of infection. One should document the presence or absence of thrombi or
vegetations in the diverse parts in contact with blood. The anastomotic sites
to the left ventricle and the aorta should be described and sampled for
histologic examination. As is true for any prosthetic device, document any
identifying numbers or labels and place the device in a “per-manent save” area.
Pacemakers and Defibrillators
Recording
the serial number of the pacemaker or defibrillator is the first step. These
numbers are usually easy to find when the fibrous sac of tissue surrounding the
device is incised. On gross examination, the important things to document are
the extent of adhesions on the leads and the location of the active interface
of these devices (i.e., the electrode tips) within the heart. Rarely, these
devices show infected vegetations. Submit representative sections of any
adherent fibrous tissue. Again, as was true for left ventricular assist
devices, the device should be placed in a “perma-nent save” area.

Important Issues to Address in Your Surgical Pathology Report on Heart Valves
Native Heart Valves
·
How many leaflets are present?
·
Do the leaflets show evidence of fusion, myxoid
change, fibrosis, calcification, thrombi, or vege-tations?
· Do the
chordae show evidence of shortening, thickening, stretching, fusion, or
rupture?
· Are the
papillary muscles infarcted?
· Do any
of these changes appear to have an impact on valve function?
Mechanical and Bioprosthetic Valves
· What
type of valve is it?
· Is there
evidence of thrombi, fibrous tissue overgrowth, wear and tear, or mechanical
failure?
· Is the
valve function compromised?
Related Topics