Chapter: Biochemistry: Storage Mechanisms and Control in Carbohydrate Metabolism
Glucose Is Sometimes Diverted throughthe Pentose Phosphate Pathway
Glucose Is Sometimes Diverted
throughthe Pentose Phosphate Pathway
The pentose phosphate
pathway is an alternative to glycolysis and differs from it in several
important ways. In glycolysis, one of our most important concerns was the
production of ATP. In the pentose phosphate pathway, the production of ATP is
not the crux of the matter. As the name of the pathway indicates, five-carbon
sugars, including ribose, are produced from glucose. Ribose and its derivative
deoxyribose play an important role in the structure of nucleic acids. Another
important facet of the pentose phosphate pathway is the production of
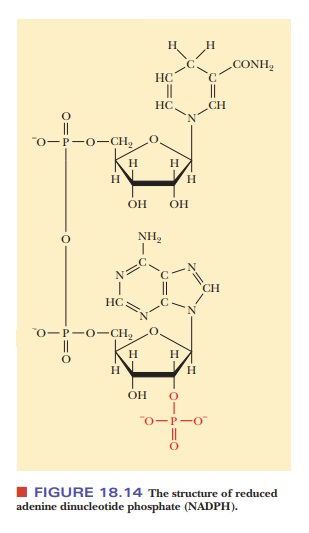
nicotinamide adenine dinucleotide phosphate (NADPH), a compound that differs
from nicotinamide adenine dinucleotide (NADH) by having one extra phosphate
group esterified to the ribose ring of the adenine nucleotide portion of the
molecule (Figure 18.14). A more important difference is the way these two
coenzymes function. NADH is produced in the oxidative reactions that give rise
to ATP. NADPH is a reducing agent in biosynthesis, which, by its very nature,
is a reductive process.
The pentose phosphate pathway begins with a series of oxidation
reactions that produce NADPH and five-carbon sugars. The remainder of the
path-way involves nonoxidative reshuffling of the carbon skeletons of the
sugars involved. The products of these nonoxidative reactions include
substances such as fructose-6-phosphate and glyceraldehyde-3-phosphate, which
play a role in glycolysis. Some of these reshuffling reactions will reappear
when we look at the production of sugars in photosynthesis.
What are the oxidative reactions of the pentose phosphate pathway?
In the first reaction of the pathway, glucose-6-phosphate is
oxidized to 6-phosphogluconate (Figure 18.15, top). The enzyme that catalyzes this reaction is glucose-6-phosphate dehydrogenase. Note
that NADPH is produced by the reaction.
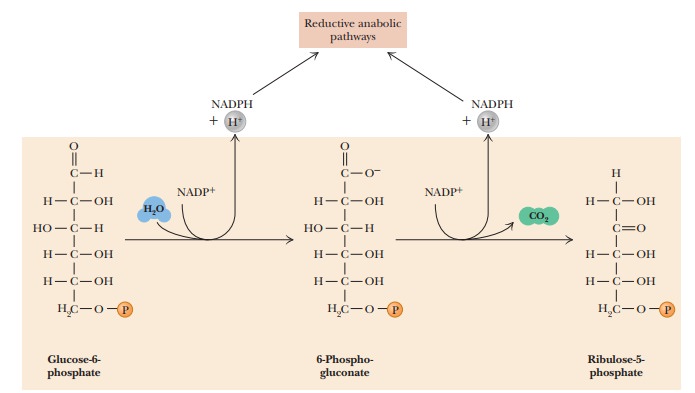
The next reaction is an oxidative decarboxylation, and NADPH is
produced once again. The 6-phosphogluconate molecule loses its carboxyl group,
which is released as carbon dioxide, and the five-carbon keto-sugar (ketose)
ribulose-5-phosphate is the other product. The enzyme that catalyzes this
reaction is 6-phosphogluconate
dehydrogenase. In the process, the C-3 hydroxyl group of
the6-phosphogluconate is oxidized to form a β-keto acid, which is unstable and readily
decarboxylates to form ribulose-5-phosphate.
What are the nonoxidative reactions of the pentose phosphate pathway, and why are they important?
In the remaining steps of the pentose phosphate pathway, several
reactions involve transfer of two- and three-carbon units. To keep track of the
carbon backbone of the sugars and their aldehyde and ketone functional groups,
we shall write the formulas in the open-chain form.
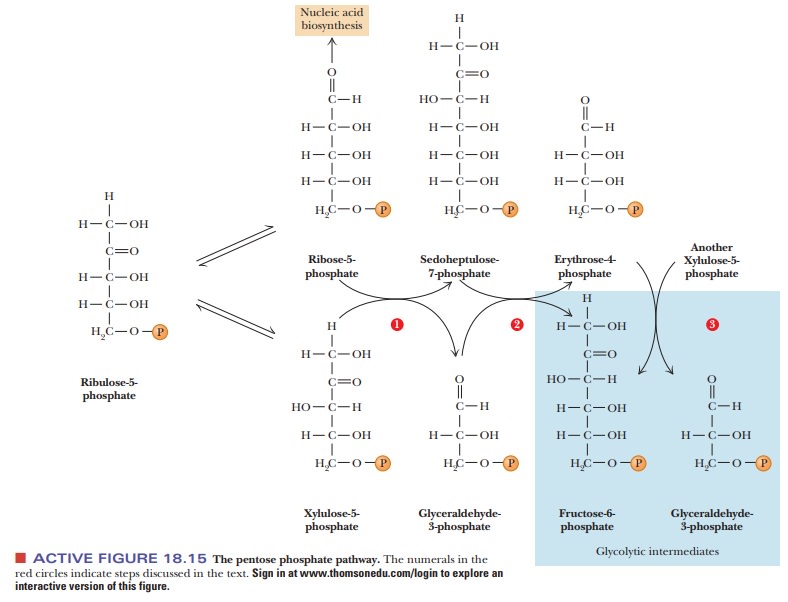
There are two different reactions in which ribulose-5-phosphate isomer-izes. In one of these reactions, catalyzed by phosphopentose-3-epimerase, there is an inversion of configuration around carbon atom 3, producing xylulose-5-phosphate, which is also a ketose (Figure 18.15, bottom). The other isomeriza-tion reaction, catalyzed by phosphopentose isomerase, produces a sugar with an aldehyde group (an aldose) rather than a ketone. In this second reaction, ribulose-5-phosphate isomerizes to ribose-5-phosphate (Figure 18.15, bottom). Ribose-5-phosphate is a necessary building block for the synthesis of nucleic acids and coenzymes such as NADH.
The group-transfer reactions that link the pentose phosphate
pathway with glycolysis require the two five-carbon sugars produced by the
isomeriza-tion of ribulose-5-phosphate. Two molecules of xylulose-5-phosphate
and one molecule of ribose-5-phosphate rearrange to give two molecules of
fructose-6-phosphate and one molecule of glyceraldehyde-3-phosphate. In other
words, three molecules of pentose (with five carbon atoms each) give two molecules
of hexose (with six carbon atoms each) and one molecule of a triose (with three
carbon atoms). The total number of carbon atoms (15) does not change, but there
is considerable rearrangement as a result of group transfer.
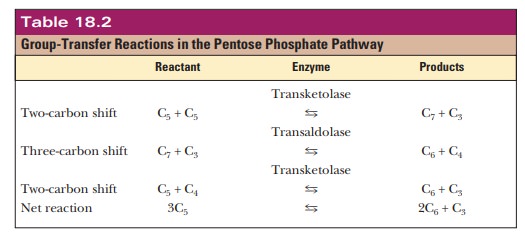
Two enzymes, transketolase
and transaldolase, are responsible
for the reshuf-fling of the carbon atoms of sugars such as ribose-5-phosphate
and xylulose-5-phosphate in the remainder of the pathway, which consists of
three reactions. Transketolase transfers a two-carbon unit. Transaldolase
transfers a three-carbon unit. Transketolase catalyzes the first and third
reactions in the rear-rangement process, and transaldolase catalyzes the second
reaction. The results of these transfers are summarized in Table 18.2. In the
first of these reactions, a two-carbon unit from xylulose-5-phosphate (five
carbons) is trans-ferred to ribose-5-phosphate (five carbons) to give
sedoheptulose-7-phosphate (seven carbons) and glyceraldehyde-3-phosphate (three
carbons), as shown in Figure 18.15, bottom,red
numeral 1.
In the reaction catalyzed by transaldolase, a three-carbon unit is
trans-ferred from the seven-carbon sedoheptulose-7-phosphate to the
three-carbon glyceraldehyde-3-phosphate (Figure 18.15, red numeral 2). The
products of the reaction are fructose-6-phosphate (six carbons) and
erythrose-4-phos-phate (four carbons).
In the final reaction of this type in the pathway,
xylulose-5-phosphate reacts with erythrose-4-phosphate. This reaction is
catalyzed by transketolase. The products of the reaction are fructose-6-phosphate
and glyceraldehyde-3-phosphate (Figure 18.15, red numeral 3).
In the pentose phosphate pathway, glucose-6-phosphate can be
converted to fructose-6-phosphate and glyceraldehyde-3-phosphate by a means
other than the glycolytic pathway. For this reason, the pentose phosphate
pathway is also called the hexose
monophosphate shunt, and this name is used in some texts. A major feature
of the pentose phosphate pathway is the production of ribose-5-phosphate and
NADPH. The control mechanisms of the pentose phosphate pathway can respond to
the varying needs of organisms for either or both of these compounds.
How is the pentose phosphate pathway controlled?
As we have seen, the reactions catalyzed by transketolase and transaldolase are reversible, which allows the pentose phosphate pathway to respond to the needs of an organism.
The starting material, glucose-6-phosphate, undergoes different
reactions depending on whether there is a greater need for ribose-5-phosphate
or for NADPH. The operation of the oxidative portion of the pathway depends
strongly on the organism’s requirement for NADPH. The need for
ribose-5-phosphate can be met in other ways, since ribose-5-phosphate can be
obtained from glycolytic intermediates without the oxidative reactions of the
pentose phosphate pathway (Figure 18.16).
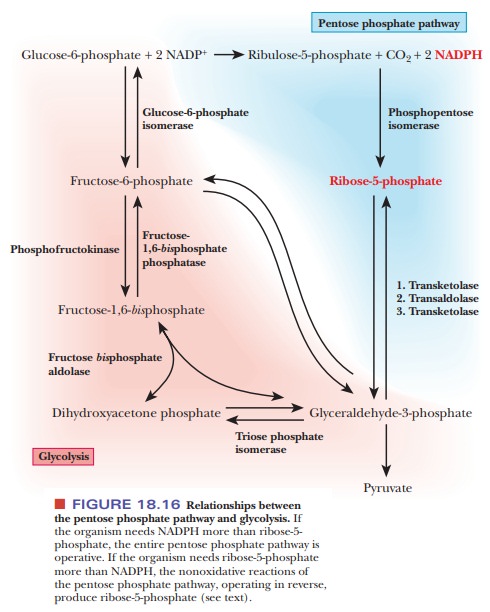
If the
organism needs more NADPH than ribose-5-phosphate, the reaction series goes
through the complete pathway just discussed. The oxidative reac-tions at the
beginning of the pathway are needed to produce NADPH. The net reaction for the
oxidative portion of the pathway is
6 Glucose-6-phosphate + 12NADP+ + 6H2O - > 6 Ribose-5-phosphate + 6CO2 + 12NADPH + 12H+
The
following Biochemical Connections box discusses a clinical manifestation of an
enzyme malfunction in the pentose phosphate pathway.
If the organism has a greater need for ribose-5-phosphate than for NADPH, fructose-6-phosphate and glyceraldehyde-3-phosphate can give rise to ribose-5-phosphate by the successive operation of the transketolase and transaldolase reactions, bypassing the oxidative portion of the pentose phosphate pathway (follow the red shaded path down to glyceraldehyde-3-phosphate and then go up to ribose-5-phosphate) (Figure 18.16). The reactions catalyzed by transketo-lase and transaldolase are reversible, and this fact plays an important role in the organism’s ability to adjust its metabolism to changes in conditions. We shall now look at the mode of action of these two enzymes.
Transaldolase
has many features in common with the enzyme aldolase, which we met in the
glycolytic pathway. Both an aldol cleavage and an aldol condensa-tion occur at
different stages of the reaction. We already saw the mechanism of aldol
cleavage, involving the formation of a Schiff base, when we discussed the
aldolase reaction in glycolysis, and we need not discuss this point further.
Transketolase resembles pyruvate decarboxylase, the enzyme that
converts pyruvate to acetaldehyde, in that it also requires Mg2+ and
thia-mine pyrophosphate (TPP). As in the pyruvate decarboxylase reaction, a
carb-anion plays a crucial role in the reaction mechanism, which is similar to
that of the conversion of pyruvate to acetaldehyde.
Summary
In the pentose phosphate pathway, two important processes take
place. One is the formation of five-carbon sugars, particularly ribose, a
compo-nent of RNA.
The other is the formation of NADPH, a reducing
agent required in many anabolic reactions.
Related Topics