Chapter: Biochemistry: Protein Purification and Characterization Techniques
Electrophoresis
Electrophoresis
Electrophoresis is based on the motion of charged particles in an electricfield toward an electrode of opposite charge. Macromolecules have differing mobilities based on their charge, shape, and size. Although many supporting media have been used for electrophoresis, including paper and liquid, the most common support is a polymer of agarose or acrylamide that is similar to those used for column chromatography.
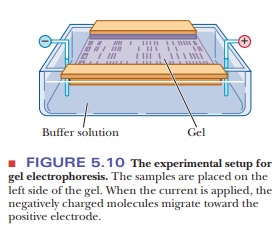
A sample is applied to wells that are formed in the
supporting medium. An electric current is passed through the medium at a
controlled voltage to achieve the desired separation (Figure 5.10). After the
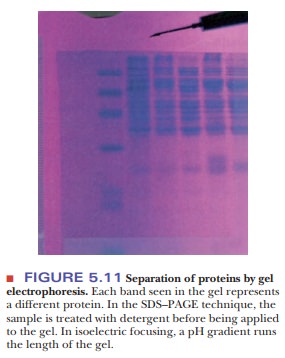
proteins are separated on the gel, the gel is stained to reveal the protein
locations, as shown in Figure 5.11.
What is the difference between agarose gels and polyacrylamide gels?
Agarose-based gels are most often used to separate nucleic acids and will be discussed. For proteins, the most common electrophoretic support is polyacrylamide (Figure 5.4), although sometimes agarose is used. A poly-acrylamide gel is prepared and cast as a continuous cross-linked matrix, rather than being produced in the bead form employed in column chromatography. In one variation of polyacrylamide-gel electrophoresis, the protein sample is treated with the detergent sodium dodecyl sulfate (SDS) before it is applied to the gel. The structure of SDS is CH3(CH2)10CH2OSO3Na+. The anion binds strongly to proteins via nonspecific adsorption. The larger the protein, the more of the anion it adsorbs. SDS completely denatures proteins, breaking all the noncovalent interactions that determine tertiary and quaternary structure. This means that multisubunit proteins can be analyzed as the component polypeptide chains. All the proteins in a sample have a negative charge as a result of adsorption of the anionic SO3–. The proteins also have roughly the same shape, which is a random coil. In SDS–polyacrylamide-gel electrophoresis(SDS–PAGE), the acrylamide offers more resistance to large molecules than tosmall molecules.
Because the shape and charge are approximately the same for all the proteins in the sample, the size of the
protein becomes the determining factor in the separation: small proteins move
faster than large ones. Like molecular-sieve chromatography, SDS–PAGE can be
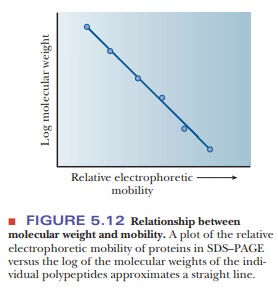
used to estimate the molecular weights of proteins by comparing the sample with
standard samples. For most proteins, the log of the molecular weight is
linearly related to its mobility on SDS–PAGE, as shown in Figure 5.12. Proteins
can also be separated on acrylamide without the SDS, in which case the gel is
called a native gel. This is
useful for times when the study calls for a protein in its native conformation.
In this case, however, the mobility is not correlated with size specifically,
as three variables control the movement down the gel-size, shape, and charge.
Isoelectric focusing is
another variation of gel electrophoresis. Because different proteins have
different titratable groups, they also have different isoelectric points.
Recall that the isoelectric pH (pI) is the pH at which a protein (or amino acid
or peptide) has no net charge. At the pI, the number of positive charges
exactly balances the number of negative charges. In an isoelectric focusing
experiment, the gel is prepared with a pH gradient that parallels the
electric-field gradient. As proteins migrate through the gel under the
influence of the electric field, they encounter regions of different pH, so the
charge on the protein changes. Eventually each protein reaches the point at
which it has no net charge-its isoelectric point-and no longer migrates. Each
protein remains at the position on the gel corresponding to its pI, allow- ing
for an effective method of separation.
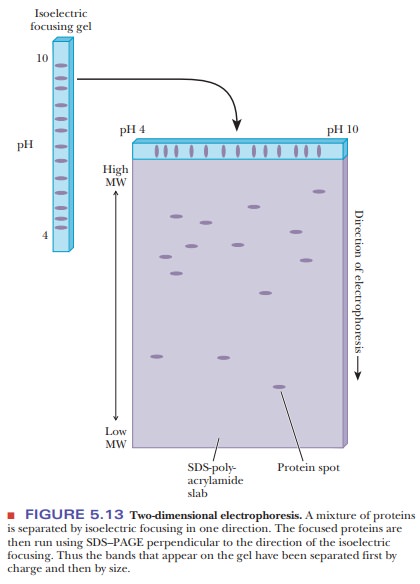
An ingenious combination, known as two-dimensional gel electrophoresis (2-D gels), allows for enhanced separation by using isoelectric focusing in one dimension and SDS–PAGE run at 90° to the first (Figure 5.13).
Summary
Electrophoresis separates molecules on a gel
medium by passing electri-cal current through the gel.
Proteins are separated on the gel based on
their size, shape, and charge.
With
SDS–polyacrylamide-gel electrophoresis, proteins separate based on molecular
weight.
Related Topics