Chapter: Medical Physiology: Insulin, Glucagon, and Diabetes Mellitus
Effect of Insulin on Carbohydrate Metabolism
Effect of Insulin on Carbohydrate Metabolism
Immediately after a high-carbohydrate meal, the glucose that is absorbed into the blood causes rapid secretion of insulin, which is discussed in detail later. The insulin in turn causes rapid uptake, storage, and use of glucose by almost all tissues of the body, but especially by the muscles, adipose tissue, and liver.
Insulin Promotes Muscle Glucose Uptake and Metabolism
During much of the day, muscle tissue depends not on glucose for its energy but on fatty acids. The principal reason for this is that the normal resting muscle mem-brane is only slightly permeable to glucose, except when the muscle fiber is stimulated by insulin; between meals, the amount of insulin that is secreted is too small to promote significant amounts of glucose entry into the muscle cells.
However, under two conditions the muscles do use large amounts of glucose. One of these is during mod-erate or heavy exercise. This usage of glucose does not require large amounts of insulin, because exercising muscle fibers become more permeable to glucose even in the absence of insulin because of the contraction process itself.
The second condition for muscle usage of large amounts of glucose is during the few hours after a meal. At this time the blood glucose concentration is high and the pancreas is secreting large quantities of insulin. The extra insulin causes rapid transport of glucose into the muscle cells. This causes the muscle cell during this period to use glucose preferentially over fatty acids, as we discuss later.
Storage of Glycogen in Muscle. If the muscles are notexercising after a meal and yet glucose is transported into the muscle cells in abundance, then most of the glucose is stored in the form of muscle glycogen instead of being used for energy, up to a limit of 2 to 3 per cent concentration. The glycogen can later be used for energy by the muscle. It is especially useful for short periods of extreme energy use by the muscles and even to provide spurts of anaerobic energy for a few minutes at a time by glycolytic breakdown of the glycogen to lactic acid, which can occur even in the absence of oxygen.
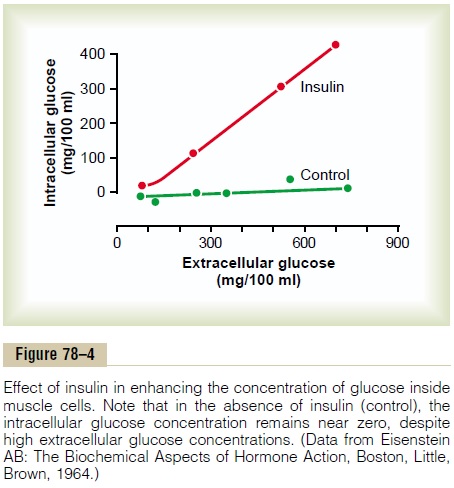
Quantitative Effect of Insulin to Facilitate Glucose Transport Through the Muscle Cell Membrane. The quantitativeeffect of insulin to facilitate glucose transport through the muscle cell membrane is demonstrated by the experimental results shown in Figure 78–4. The lower curve labeled “control” shows the concentration of free glucose measured inside the cell, demonstrating that the glucose concentration remained almost zero despite increased extracellular glucose concentration up to as high as 750 mg/100 ml. In contrast, the curve labeled “insulin” demonstrates that the intracellular glucose concentration rose to as high as 400 mg/100 ml when insulin was added. Thus, it is clear that insulin can increase the rate of transport of glucose into the resting muscle cell by at least 15-fold.
Insulin Promotes Liver Uptake, Storage, and Use of Glucose
One of the most important of all the effects of insulin is to cause most of the glucose absorbed after a meal to be stored almost immediately in the liver in the form of glycogen. Then, between meals, when food is not available and the blood glucose concentration begins to fall, insulin secretion decreases rapidly and the liver glycogen is split back into glucose, which is released back into the blood to keep the glucose con-centration from falling too low.
The mechanism by which insulin causes glucose uptake and storage in the liver includes several almost simultaneous steps:
1. Insulin inactivates liver phosphorylase, the principal enzyme that causes liver glycogen to split into glucose. This prevents breakdown of the glycogen that has been stored in the liver cells.
2. Insulin causes enhanced uptake of glucose from the blood by the liver cells. It does this by increasing the activity of the enzyme glucokinase,which is one of the enzymes that causes the initial phosphorylation of glucose after it diffuses into the liver cells. Once phosphorylated, the glucose istemporarily trapped inside the liver cells becausephosphorylated glucose cannot diffuse back through the cell membrane.
3. Insulin also increases the activities of the enzymes that promote glycogen synthesis, including especially glycogen synthase, which is responsible for polymerization of the monosaccharide units to form the glycogen molecules.
The net effect of all these actions is to increase the amount of glycogen in the liver. The glycogen can increase to a total of about 5 to 6 per cent of the liver mass, which is equivalent to almost 100 grams of stored glycogen in the whole liver.
Glucose Is Released from the Liver Between Meals. When theblood glucose level begins to fall to a low level between meals, several events transpire that cause the liver to release glucose back into the circulating blood:
1. The decreasing blood glucose causes the pancreas to decrease its insulin secretion.
2. The lack of insulin then reverses all the effects listed earlier for glycogen storage, essentially stopping further synthesis of glycogen in the liver and preventing further uptake of glucose by the liver from the blood.
3. The lack of insulin (along with increase of glucagon, which is discussed later) activates the enzyme phosphorylase, which causes the splitting of glycogen into glucose phosphate.
4. The enzyme glucose phosphatase, which had been inhibited by insulin, now becomes activated by the insulin lack and causes the phosphate radical to split away from the glucose; this allows the free glucose to diffuse back into the blood.
Thus, the liver removes glucose from the blood when it is present in excess after a meal and returns it to the blood when the blood glucose concentration falls between meals. Ordinarily, about 60 per cent of the glucose in the meal is stored in this way in the liver and then returned later.
Insulin Promotes Conversion of Excess Glucose into Fatty Acids and Inhibits Gluconeogenesis in the Liver. When the quan-tity of glucose entering the liver cells is more than can be stored as glycogen or can be used for local hepato-cyte metabolism, insulin promotes the conversionof all this excess glucose into fatty acids. These fattyacids are subsequently packaged as triglycerides in very-low-density lipoproteins and transported in this form by way of the blood to the adipose tissue and deposited as fat.
Insulin also inhibits gluconeogenesis. It does this mainly by decreasing the quantities and activities of the liver enzymes required for gluconeogenesis. However, part of the effect is caused by an action of insulin that decreases the release of amino acids from muscle and other extrahepatic tissues and in turn the availability of these necessary precursors required for gluconeogenesis. This is discussed further in relation to the effect of insulin on protein metabolism.
Lack of Effect of Insulin on Glucose Uptake and Usage by the Brain
The brain is quite different from most other tissues of the body in that insulin has little effect on uptake or use of glucose. Instead, the brain cells are permeable toglucose and can use glucose without the intermediation of insulin.
The brain cells are also quite different from most other cells of the body in that they normally use only glucose for energy and can use other energy sub-strates, such as fats, only with difficulty. Therefore, it is essential that the blood glucose level always be main-tained above a critical level, which is one of the most important functions of the blood glucose control system. When the blood glucose falls too low, into the range of 20 to 50 mg/100 ml, symptoms of hypo-glycemic shock develop, characterized by progressivenervous irritability that leads to fainting, seizures, and even coma.
Effect of Insulin on Carbohydrate Metabolism in Other Cells
Insulin increases glucose transport into and glucose usage by most other cells of the body (with the excep-tion of the brain cells, as noted) in the same way that it affects glucose transport and usage in muscle cells. The transport of glucose into adipose cells mainly pro-vides substrate for the glycerol portion of the fat mol-ecule. Therefore, in this indirect way, insulin promotes deposition of fat in these cells.
Related Topics