Chapter: Medical Physiology: Membrane Physiology, Nerve, and Muscle : Transport of Substances Through the Cell Membrane
Diffusion Through Protein Channels, and ÔÇťGatingÔÇŁ of These Channels
Diffusion Through Protein Channels, and ÔÇťGatingÔÇŁ of These Channels
Computerized three-dimensional reconstructions of protein channels have demonstrated tubular pathways all the way from the extracellular to the intracellular fluid. Therefore, substances can move by simple diffusion directly along these channels from one side of the membrane to the other. The protein channels are distinguished by two important characteristics: (1) they are often selectively permeable to certain sub-stances, and (2) many of the channels can be opened or closed by gates.
Selective Permeability of Protein Channels. Many of theprotein channels are highly selective for transport of one or more specific ions or molecules. This results from the characteristics of the channel itself, such as its diameter, its shape, and the nature of the electrical charges and chemical bonds along its inside surfaces. To give an example, one of the most important of the protein channels, the so-called sodium channel, is only 0.3 by 0.5 nanometer in diameter, but more important, the inner surfaces of this channel are strongly nega-tively charged, as shown by the negative signs insidethe channel proteins in the top panel of Figure 4ÔÇô4.
These strong negative charges can pull small dehy-drated sodium ions into these channels, actually pullingthe sodium ions away from their hydrating water molecules. Once in the channel, the sodium ions diffuse in either direction according to the usual laws of diffusion. Thus, the sodium channel is specifically selective for passage of sodium ions.
Conversely, another set of protein channels is selec-tive for potassium transport, shown in the lower panel of Figure 4ÔÇô4. These channels are slightly smaller than the sodium channels, only 0.3 by 0.3 nanometer, but they are not negatively charged, and their chemicalbonds are different. Therefore, no strong attrac-tive force is pulling ions into the channels, and the potassium ions are not pulled away from the water
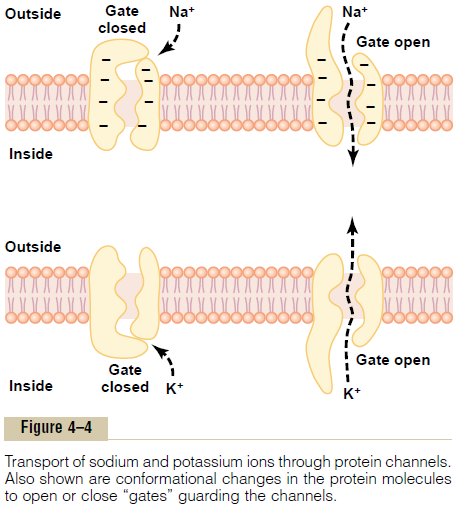
molecules that hydrate them. The hydrated form of the potassium ion is considerably smaller than the hydrated form of sodium because the sodium ion attracts far more water molecules than does potas-sium. Therefore, the smaller hydrated potassium ions can pass easily through this small channel, whereas the larger hydrated sodium ions are rejected, thus provid-ing selective permeability for a specific ion.
Gating of Protein Channels. Gating of protein channelsprovides a means of controlling ion permeability of the channels. This is shown in both panels of Figure 4ÔÇô4 for selective gating of sodium and potassium ions. It is believed that some of the gates are actual gatelike extensions of the transport protein molecule, which can close the opening of the channel or can be lifted away from the opening by a conformational change in the shape of the protein molecule itself.
The opening and closing of gates are controlled in two principal ways:
1. Voltage gating. In this instance, the molecularconformation of the gate or of its chemical bonds responds to the electrical potential across the cell membrane. For instance, in the top panel of Figure 4ÔÇô4, when there is a strong negative charge on the inside of the cell membrane, this presumably could cause the outside sodium gates to remain tightly closed; conversely, when the inside of the membrane loses its negative charge, these gates would open suddenly and allow tremendous quantities of sodium to pass inward through the sodium pores. This is the basic mechanism for eliciting action potentials in nerves that are responsible for nerve signals. In the bottom panel of Figure 4ÔÇô4, the potassium gates are on the intracellular ends of the potassium channels, and they open when the inside of the cell membrane becomes positively charged. The opening of these gates is partly responsible for terminating the action potential.
2. Chemical (ligand) gating. Some protein channelgates are opened by the binding of a chemical substance (a ligand) with the protein; this causes a conformational or chemical bonding change in the protein molecule that opens or closes the gate. This is calledchemical gating or ligand gating. One of the most important instances of chemical gating is the effect of acetylcholine on the so-called acetylcholine channel. Acetylcholine opens the gateof this channel, providing a negatively charged pore about 0.65 nanometer in diameter that allows uncharged molecules or positive ions smaller than this diameter to pass through. This gate is exceedingly important for the transmission of nerve signals from one nerve cell to another and from nerve cells to muscle cells to cause muscle contraction.
Open-State Versus Closed-State of Gated Channels.
Figure 4ÔÇô5A shows an especially interesting charac-teristic of most voltage-gated channels. This figure shows two recordings of electrical current flowing through a single sodium channel when there was an approximate 25-millivolt potential gradient across the
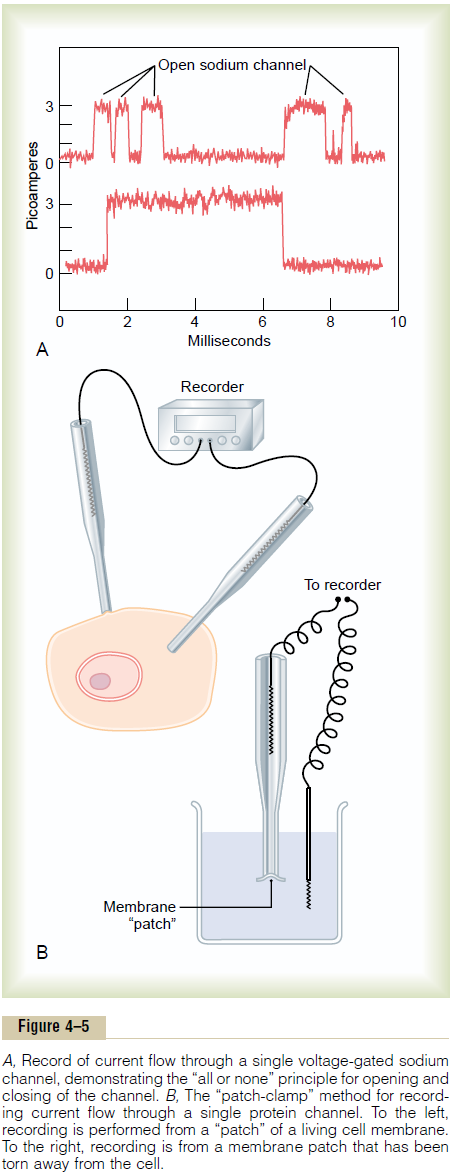
membrane. Note that the channel conducts current either ÔÇťall or none.ÔÇŁ That is, the gate of the channel snaps open and then snaps closed, each open state lasting for only a fraction of a millisecond up to several milliseconds. This demonstrates the rapidity with which changes can occur during the opening and closing of the protein molecular gates. At one voltage potential, the channel may remain closed all the time or almost all the time, whereas at another voltage level, it may remain open either all or most of the time. At in-between voltages, as shown in the figure, the gates tend to snap open and closed intermittently, giving an average current flow somewhere between the minimum and the maximum.
Patch-Clamp Method for Recording Ion Current Flow Through Single Channels.
One might wonder how it is technicallypossible to record ion current flow through single protein channels as shown in Figure 4ÔÇô5A.This has been achieved by using the ÔÇťpatch-clampÔÇŁ method illustrated in Figure 4ÔÇô5B. Very simply, a micropipette, having a tip diameter of only 1 or 2 micrometers, is abutted against the outside of a cell membrane. Then suction is applied inside the pipette to pull the membrane against the tip of the pipette. This creates a seal where the edges of the pipette touch the cell membrane. The result is a minute membrane ÔÇťpatchÔÇŁ at the tip of the pipette through which electrical current flow can be recorded.
Alternatively, as shown to the right in Figure 4ÔÇô5B, the small cell membrane patch at the end of the pipette can be torn away from the cell. The pipette with its sealed patch is then inserted into a free solution. This allows the concentrations of ions both inside the micropipette and in the outside solution to be altered as desired. Also, the voltage between the two sides of the membrane can be set at willÔÇöthat is, ÔÇťclampedÔÇŁ to a given voltage.
It has been possible to make such patches small enough so that only a single channel protein is found in the membrane patch being studied. By varying the concentrations of different ions, as well as the voltage across the membrane, one can determine the transport characteristics of the single channel and also its gating properties.
Related Topics