Chapter: Medical Physiology: Membrane Physiology, Nerve, and Muscle : Transport of Substances Through the Cell Membrane
тАЬActive TransportтАЭ of Substances Through Membranes
тАЬActive TransportтАЭ of Substances Through Membranes
At times, a large concentration of a substance is required in the intracellular fluid even though the extracellular fluid contains only a small concentration. This is true, for instance, for potassium ions. Con-versely, it is important to keep the concentrations of other ions very low inside the cell even though their concentrations in the extracellular fluid are great. This is especially true for sodium ions. Neither of these two effects could occur by simple diffusion, because simple diffusion eventually equilibrates concentrations on the two sides of the membrane. Instead, some energy source must cause excess movement of potassium ions to the inside of cells and excess movement of sodium ions to the outside of cells. When a cell membrane moves molecules or ions тАЬuphillтАЭ against a concentra-tion gradient (or тАЬuphillтАЭ against an electrical or pres-sure gradient), the process is called active transport.
Different substances that are actively transported through at least some cell membranes include sodium ions, potassium ions, calcium ions, iron ions, hydrogen ions, chloride ions, iodide ions, urate ions, several dif-ferent sugars, and most of the amino acids.
Primary Active Transport and Secondary Active Transport.
Active transport is divided into two types according tothe source of the energy used to cause the transport:primary active transport and secondary active transport.In primary active transport, the energy is deriveddirectly from breakdown of adenosine triphosphate(ATP) or of some other high-energy phosphate compound.In secondary active transport, the energy isderived secondarily from energy that has been storedin the form of ionic concentration differences ofsecondary molecular or ionic substances between thetwo sides of a cell membrane, created originally byprimary active transport. In both instances, transportdepends on carrier proteins that penetrate through thecell membrane, as is true for facilitated diffusion.However, in active transport, the carrier protein functionsdifferently from the carrier in facilitated diffusionbecause it is capable of imparting energy to thetransported substance to move it against the electrochemicalgradient. Following are some examplesof primary active transport and secondary activetransport, with more detailed explanations of theirprinciples of function.
Primary Active Transport
Sodium-Potassium Pump
Among the substances that are transported by primaryactive transport are sodium, potassium, calcium, hydrogen, chloride, and a few other ions. The active transport mechanism that has been studied in greatest detail is the sodium-potassium (Na+-K+) pump, a transport process that pumps sodium ions outward through the cell membrane of all cells and at the same time pumps potassium ions from the outside to the inside. This pump is responsible for maintaining the sodium and potassium concentration differences across the cell membrane, as well as for establishing a negative electrical voltage inside the cells.
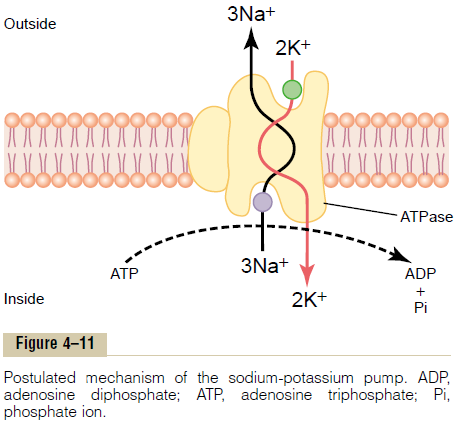
Figure 4тАУ11 shows the basic physical components of the Na+-K+ pump. The carrier protein is a complex of two separate globular proteins: a larger one called the a subunit, with a molecular weight of about 100,000, and a smaller one called the b subunit, with a molecular weight of about 55,000. Although the function of the smaller protein is not known (except that it might anchor the protein complex in the lipid membrane), the larger protein has three specific features that are important for the functioning of the pump:
1. It has three receptor sites for binding sodium ions on the portion of the protein that protrudes to the inside of the cell.
2. It has two receptor sites for potassium ions on the outside.
3. The inside portion of this protein near the sodium binding sites has ATPase activity.
To put the pump into perspective:When two potassium ions bind on the outside of the carrier protein
and three sodium ions bind on the inside, the ATPasefunction of the protein becomes activated. This thencleaves one molecule of ATP, splitting it to adenosinediphosphate (ADP) and liberating a high-energyphosphate bond of energy. This liberated energy isthen believed to cause a chemical and conformationalchange in the protein carrier molecule, extruding thethree sodium ions to the outside and the two potassiumions to the inside.
As with other enzymes, the Na+-K+ ATPase pumpcan run in reverse. If the electrochemical gradients forNa+ and K+ are experimentally increased enough sothat the energy stored in their gradients is greater thanthe chemical energy of ATP hydrolysis, these ions willmove down their concentration gradients and the Na+-K+ pump will synthesize ATP from ADP and phosphate.The phosphorylated form of the Na+-K+ pump,therefore, can either donate its phosphate to ADP toproduce ATP or use the energy to change its conformationand pump Na+ out of the cell and K+ into thecell. The relative concentrations of ATP, ADP, andphosphate, as well as the electrochemical gradients forNa+ and K+, determine the direction of the enzymereaction. For some cells, such as electrically activenerve cells, 60 to 70 per cent of the cellsтАЩ energyrequirement may be devoted to pumping Na+ out ofthe cell and K+ into the cell.
Importance of the Na+-K+ Pump for Controlling Cell Volume.
One of the most important functions of the Na+-K+pump is to control the volume of each cell. Withoutfunction of this pump, most cells of the body wouldswell until they burst. The mechanism for controllingthe volume is as follows: Inside the cell are largenumbers of proteins and other organic molecules thatcannot escape from the cell. Most of these are negativelycharged and therefore attract large numbers ofpotassium, sodium, and other positive ions as well. Allthese molecules and ions then cause osmosis of waterto the interior of the cell. Unless this is checked, thecell will swell indefinitely until it bursts. The normal mechanism for preventing this is the Na+-K+ pump. Note again that this device pumps three Na+ ions to the outside of the cell for every two K+ ions pumped to the interior. Also, the membrane is far less perme-able to sodium ions than to potassium ions, so that once the sodium ions are on the outside, they have a strong tendency to stay there. Thus, this represents a net loss of ions out of the cell, which initiates osmosis of water out of the cell as well.
If a cell begins to swell for any reason, this auto-matically activates the Na+-K+ pump, moving still more ions to the exterior and carrying water with them. Therefore, the Na+-K+ pump performs a continual sur-veillance role in maintaining normal cell volume.
Electrogenic Nature of the Na+-K+ Pump. The fact that theNa+-K+ pump moves three Na+ ions to the exterior for every two K+ ions to the interior means that a net of one positive charge is moved from the interior of the cell to the exterior for each cycle of the pump. This creates pos-itivity outside the cell but leaves a deficit of positive ions inside the cell; that is, it causes negativity on the inside. Therefore, the Na+-K+ pump is said to beelectrogenic because it creates an electrical potential across the cell membrane. As discussed, this electrical potential is a basic requirement in nerve and muscle fibers for transmitting nerve and muscle signals.
Primary Active Transport of Calcium Ions
Another important primary active transport mecha-nism is the calcium pump. Calcium ions are normally maintained at extremely low concentration in the intracellular cytosol of virtually all cells in the body, at a concentration about 10,000 times less than that in the extracellular fluid. This is achieved mainly by two primary active transport calcium pumps. One is in the cell membrane and pumps calcium to the outside of the cell. The other pumps calcium ions into one or more of the intracellular vesicular organelles of the cell, such as the sarcoplasmic reticulum of muscle cells and the mitochondria in all cells. In each of these instances, the carrier protein penetrates the membrane and functions as an enzyme ATPase, having the same capability to cleave ATP as the ATPase of the sodium carrier protein. The difference is that this protein has a highly specific binding site for calcium instead of for sodium.
Primary Active Transport of Hydrogen Ions
At two places in the body, primary active transport of hydrogen ions is very important: (1) in the gastric glands of the stomach, and (2) in the late distal tubules and cortical collecting ducts of the kidneys.
In the gastric glands, the deep-lying parietal cells have the most potent primary active mechanism for transporting hydrogen ions of any part of the body. This is the basis for secreting hydrochloric acid in the stomach digestive secretions. At the secretory ends of the gastric gland parietal cells, the hydrogen ion con-centration is increased as much as a millionfold and then released into the stomach along with chloride ions to form hydrochloric acid.
In the renal tubules are special intercalated cells in the late distal tubules and cortical collecting ducts that also transport hydrogen ions by primary active trans-port. In this case, large amounts of hydrogen ions are secreted from the blood into the urine for the purpose of eliminating excess hydrogen ions from the body fluids. The hydrogen ions can be secreted into the urine against a concentration gradient of about 900-fold.
Energetics of Primary Active Transport
The amount of energy required to transport a sub-stance actively through a membrane is determined by how much the substance is concentrated during trans-port. Compared with the energy required to concen-trate a substance 10-fold, to concentrate it 100-fold requires twice as much energy, and to concentrate it 1000-fold requires three times as much energy. In other words, the energy required is proportional to the logarithm of the degree that the substance is concentrated, as expressed by the following formula:

Thus, in terms of calories, the amount of energy required to concentrate 1 osmole of substance 10-fold is about 1400 calories; or to concentrate it 100-fold, 2800 calories. One can see that the energy expenditure for concentrating substances in cells or for removing substances from cells against a concentration gradient can be tremendous. Some cells, such as those lining the renal tubules and many glandular cells, expend as much as 90 per cent of their energy for this purpose alone.
Secondary Active Transport - Co-Transport and Counter-Transport
When sodium ions are transported out of cells by primary active transport, a large concentration gradi-ent of sodium ions across the cell membrane usually developsтАФhigh concentration outside the cell and very low concentration inside. This gradient represents a storehouse of energy because the excess sodium outside the cell membrane is always attempting to diffuse to the interior. Under appropriate conditions, this diffusion energy of sodium can pull other sub-stances along with the sodium through the cell mem-brane. This phenomenon is called co-transport; it is one form of secondary active transport.
For sodium to pull another substance along with it, a coupling mechanism is required. This is achieved by means of still another carrier protein in the cell mem-brane. The carrier in this instance serves as an attach-ment point for both the sodium ion and the substance to be co-transported. Once they both are attached, the energy gradient of the sodium ion causes both the sodium ion and the other substance to be transported together to the interior of the cell.
In counter-transport, sodium ions again attempt to diffuse to the interior of the cell because of their large concentration gradient. However, this time, the sub-stance to be transported is on the inside of the cell and must be transported to the outside. Therefore, the sodium ion binds to the carrier protein where it pro-jects to the exterior surface of the membrane, while the substance to be counter-transported binds to the interior projection of the carrier protein. Once both have bound, a conformational change occurs, and energy released by the sodium ion moving to the interior causes the other substance to move to the exterior.
Co-Transport of Glucose and Amino Acids Along with Sodium Ions
Glucose and many amino acids are transported into most cells against large concentration gradients; the mechanism of this is entirely by co-transport, as shown in Figure 4тАУ12. Note that the transport carrier protein has two binding sites on its exterior side, one for sodium and one for glucose. Also, the concentration of sodium ions is very high on the outside and very low inside, which provides energy for the transport. A special property of the transport protein is that a con-formational change to allow sodium movement to the interior will not occur until a glucose molecule also attaches. When they both become attached, the con-formational change takes place automatically, and the sodium and glucose are transported to the inside of the cell at the same time. Hence, this is a sodium-glucoseco-transport mechanism.
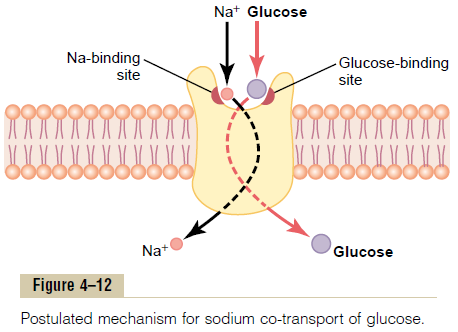
Sodium co-transport of the amino acids occurs in thesame manner as for glucose, except that it uses a dif-ferent set of transport proteins. Five amino acid trans-port proteins have been identified, each of which isresponsible for transporting one subset of amino acids with specific molecular characteristics.
Sodium co-transport of glucose and amino acids occurs especially through the epithelial cells of the intestinal tract and the renal tubules of the kidneys to promote absorption of these substances into the blood.
Other important co-transport mechanisms in at least some cells include co-transport of chloride ions, iodine ions, iron ions, and urate ions.
Sodium Counter-Transport of Calcium and Hydrogen Ions
Two especially important counter-transport mecha-nisms (transport in a direction opposite to the primary ion) are sodium-calcium counter-transport andsodium-hydrogen counter-transport.
Sodium-calcium counter-transport occurs through all or almost all cell membranes, with sodium ions moving to the interior and calcium ions to the exterior, both bound to the same transport protein in a counter-transport mode. This is in addition to primary active transport of calcium that occurs in some cells.
Sodium-hydrogen counter-transport occurs in several tissues. An especially important example is in the proximal tubules of the kidneys, where sodium ions move from the lumen of the tubule to the interior of the tubular cell, while hydrogen ions are counter-transported into the tubule lumen. As a mechanism for concentrating hydrogen ions, counter-transport is not nearly as powerful as the primary active transport of hydrogen ions that occurs in the more distal renal tubules, but it can transport extremely large numbersof hydrogen ions, thus making it a key to hydrogenion control in the body fluids.
Active Transport Through Cellular Sheets
At many places in the body, substances must be trans-ported all the way through a cellular sheet instead of simply through the cell membrane. Transport of this type occurs through the (1) intestinal epithelium, (2) epithelium of the renal tubules, (3) epithelium of all exocrine glands, (4) epithelium of the gallbladder, and (5) membrane of the choroid plexus of the brain and other membranes.
The basic mechanism for transport of a substance through a cellular sheet is (1) active transport through the cell membrane on one side of the transporting cells in the sheet, and then (2) either simple diffusion or facilitated diffusion through the membrane on the opposite side of the cell.
Figure 4тАУ13 shows a mechanism for transport of sodium ions through the epithelial sheet of the intestines, gallbladder, and renal tubules. This figure shows that the epithelial cells are connected together tightly at the luminal pole by means of junctions called тАЬkisses.тАЭ The brush border on the luminal surfaces of the cells is permeable to both sodium ions and water.
Therefore, sodium and water diffuse readily from the lumen into the interior of the cell. Then, at the basal and lateral membranes of the cells, sodium ions are actively transported into the extracellular fluid of the surrounding connective tissue and blood vessels.
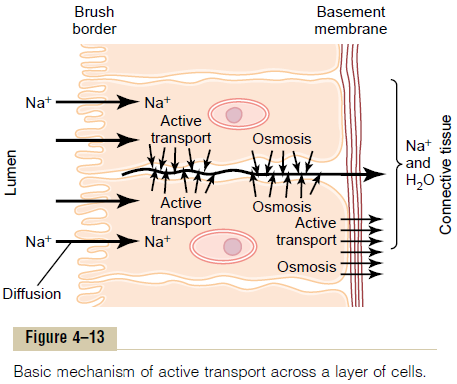
This creates a high sodium ion concentration gradient across these membranes, which in turn causes osmosis of water as well. Thus, active transport of sodium ions at the basolateral sides of the epithelial cells results in transport not only of sodium ions but also of water.
These are the mechanisms by which almost all the nutrients, ions, and other substances are absorbed into the blood from the intestine; they are also the way the same substances are reabsorbed from the glomerular filtrate by the renal tubules.
Related Topics