Chapter: Biotechnology Applying the Genetic Revolution: Viral and Prion Infections
Detection of Pathogenic Prions
DETECTION
OF PATHOGENIC PRIONS
The emergence of mad cow disease (BSE)
has created the need to screen cows and their products for the presence of the
pathogenic form of the prion protein (PrP Sc ). This is presently done by
immunological detection. Unfortunately, even though the 3D folding of the
normal (PrP c ) and pathogenic (PrP Sc ) forms is different, it has not yet
proven possible to obtain antibodies specific to each form. Because the
pathogenic form of the prion is protease resistant, samples are first treated
with protease to destroy the normal (PrP c ) form and then subjected to
immunological testing by Western blotting. The overall procedure is tedious and
of only moderate sensitivity. In particular, it would be valuable to have a
test that reveals prion disease well before symptoms develop to allow time for possible
treatments.
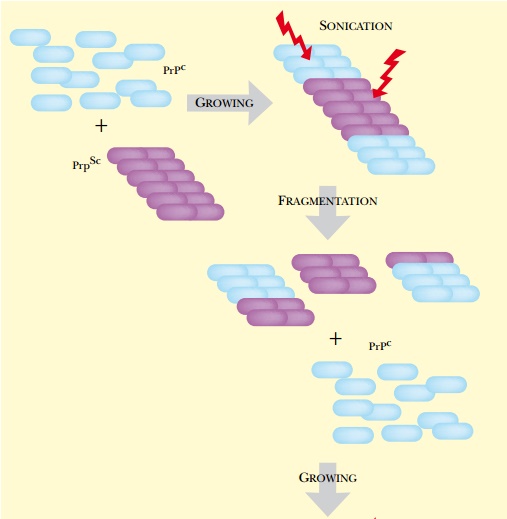
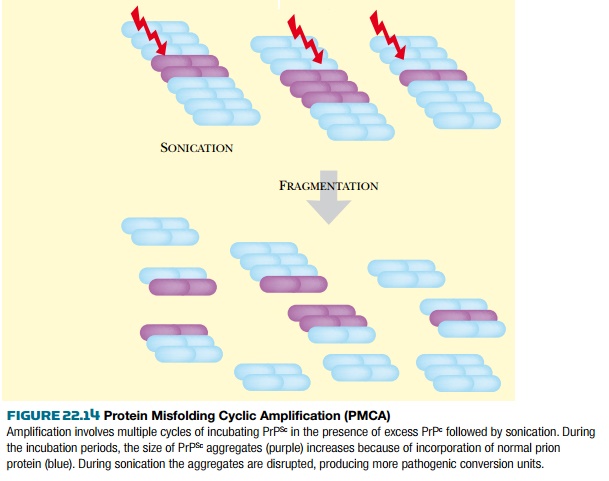
The protein misfolding cyclic
amplification (PMCA) procedure amplifies the levels of misfolded prion in a
manner analogous to the use of PCR for amplifying DNA ( Fig. 22.14 ). This
allows greatly increased sensitivity of detection of PrP Sc in clinical
samples. Small samples suspected of containing PrP Sc are mixed with normal
brain homogenate containing a surplus of the normal PrP c . The PrP c is
converted to PrP Sc and incorporated into the growing PrP Sc aggregates. The
sample is then sonicated to break up the aggregates. This procedure is repeated
for several cycles. Increases of around 60-fold over five cycles are typical.
Related Topics