Chapter: 9th Science : Heat
Concept of temperature
Concept
of temperature
Temperature is the
degree of hotness or coolness of a body. The hotter the body is higher is its
temperature.
1. Unit of Temperature
The SI unit of
temperature is kelvin (K). For day to day applications, Celsius
(˚C) is used. Temperature is measured with a thermometer.
2. Temperature scales
There are three scales
of temperature.
i.
Fahrenheit scale
ii.
Celsius or Centigrade scale
iii.
Kelvin or Absolute scale
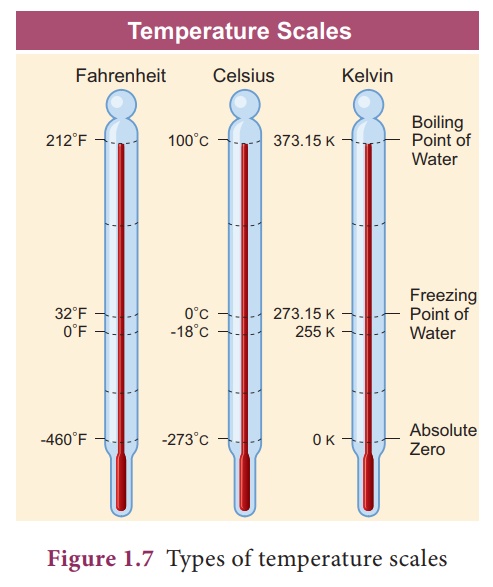
i. Fahrenheit scale
In Fahrenheit scale, 32
˚F and 212 ˚F are the freezing point and boiling points respectively. Interval
has been divided into 180 parts.
ii. Celsius temperature scale
In Celsius scale, also
called centigrade scale, 0˚C and 100 ˚C are the freezing point and boiling
respectively. Interval has been divided into 100 parts. The formula for
converting a Celsius scale to Fahrenheit scale is:
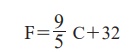
The formula for converting
a Fahrenheit scale to Celsius scale is:
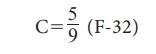
iii. Kelvin scale (Absolute scale)
Kelvin scale is known as
the absolute scale. On the Kelvin scale 0 K represents absolute zero, the
temperature at which the molecules of a substance have their lowest possible
energy. The solid, liquid, gaseous phases of water can coexist in equilibrium
at 273.16 K.
Kelvin is defined as
1/273.16 of the triple point temperature.
The formula for
converting a Celsius scale to Kelvin scale is:
K = C + 273.15
The formula for
converting a Kelvin scale to Celsius scale is:
C = K - 273.15
Absolute zero
The temperature at which
the pressure and volume of a gas theoretically reaches zero is called absolute
zero. This is shown in Figure 1.8.
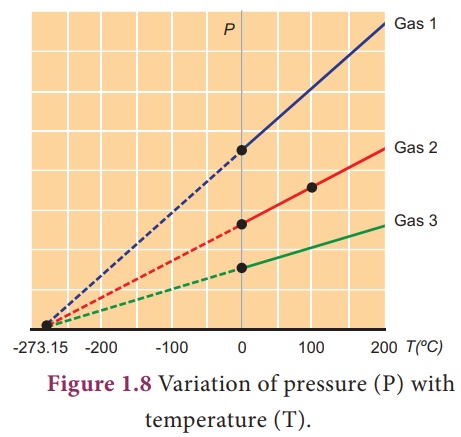
For all gases, the
pressure extrapolates to zero at the temperature -273.15 ˚C. It is known as
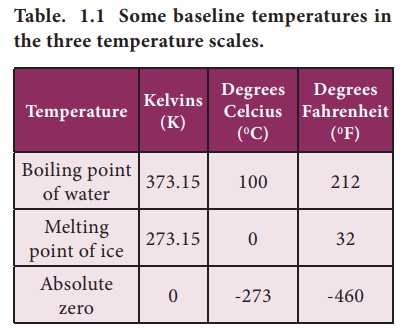
absolute zero or 0 K. Some base line temperatures in the three temperature
scales are shown in Table 1.1.
Exercise 1.1
Convert the following
i. 25 ºC to Kelvin
ii. 200 K to ºC
Solution:
i. (TK) = (T°C)
+ 273.15
(TK) = 25 +
273.15 = 298.15 K
ii. (T°C) =
(TK) - 273.15
(T°C) = 200 - 273.15 = -73.15 ºC
Exercise 1.2
Convert the following
i. 35°C to Fahrenheit
(°F)
ii. 14° F to °C
Solution:
i. T(°F) = T(°C) × 1.8 +
32
T(°F) = 25° C × 1.8 + 32
= 77° F
ii. T(°C) = (T(°F) –
32)/ 1.8
T(°C) = (14°F – 32)/ 1.8
= -10° C
Related Topics