Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Cryptococcus, Histoplasma, Coccidioides, and Other Systemic Fungal Pathogens
Cocci Dioidomycosis
COCCI DIOIDOMYCOSIS
Acute primary infection with C. immitis is either asymptomatic or presents as a complex called valley fever by residents of the endemic areas. Valley fever in- cludes fever, malaise, dry cough, joint pains, and sometimes a rash. There are few physical or radiologic findings, but the illness persists for weeks. Disseminated disease involves lesions in the bones, joints, skin, and a progressive chronic meningitis.
EPIDEMIOLOGY
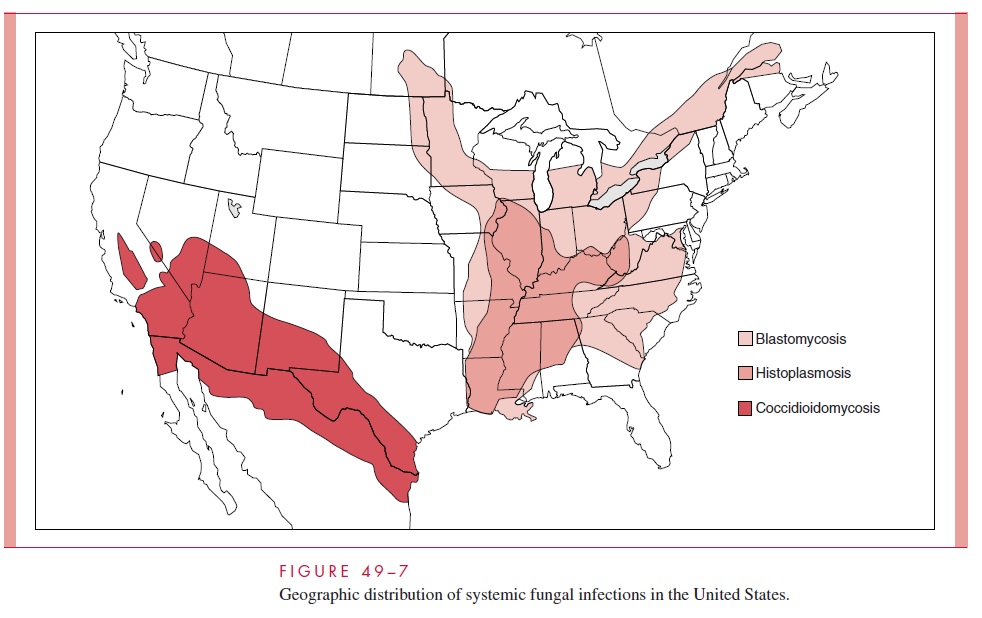
Coccidioidomycosis is the most geographically restricted of the systemic mycoses, be-cause C. immitis grows only in the alkaline soil of semiarid climates known as the Lower Sonoran life zone (see Fig 49–7). These areas are characterized by hot, dry summers, mild winters with few freezes, and annual rainfall of about 10 inches during brief rainy seasons. Areas with these conditions are found scattered throughout the Americas, some as ecologic “islands.” The primary endemic zones in the United States are in Arizona, Nevada, New Mexico, western Texas, and the arid parts of central and southern California. Persons living in the endemic areas are at high risk of infection, although disease is much less common. Positive skin test rates of 50 to 90% occur in longtime residents of highly endemic areas. Coccidioidomycosis is not transmissible from person to person.
Infection cannot be acquired without at least visiting an endemic area, although some interesting examples of the endemic zone itself paying a visit have been recorded. One such anecdote involves a gas station attendant with coccidioidomycosis whose only con-tact with an endemic area was changing a flat tire on a truck from California. In 1978, a storm originating in Bakersfield, California (endemic zone) carried a thick coat of dust all the way to San Francisco. This was followed by cases of coccidioidomycosis in persons who had never left the Bay Area. In 1992, a tenfold increase in disease in California fol-lowed an unusually wet winter in which the storms created a drought–rain–drought pat-tern just the right for growth of the mold (and wildflowers). When the Sonoran desert blooms, an arthroconidium “crop” is not far behind.
PATHOGENESIS
Inhaled arthroconidia are small enough (2 to 6μm) to bypass the defenses of the upper tracheobronchial tree and lodge in the terminal bronchioles. Human monocytes can ingest and kill some arthroconidia on initial exposure, although the outer portion of the wall of the arthroconidium has antiphagocytic properties, which persist in the early stages of spherule development. Surviving arthroconidia convert to the spherule stage, which be-gins its slow growth to a size that makes effective phagocytosis difficult. Although PMNs are able to digest the spherule wall, their access appears to be restricted by the extracellu-lar matrix surrounding it. The young endospores are released in packets that include the extracellular matrix derived from the parent spherule, which may protect them until they develop into new spherules.
A number of proteases found in the conidial cell wall or in spherules have been pro-posed as C. immitis virulence factors. In addition to their role in the fungal life cycle, some of these enzymes attack host substrates such as collagen, elastin, and immunoglob-ulins, but no direct specific contribution to disease has been defined. Components of the spherule outer wall (SOW) have been linked to virulence in animals and to strong hu-moral and cellular immune responses in humans.
IMMUNITY
Lifelong immunity to coccidioidomycosis clearly develops in the vast majority of those who become infected. This immunity is associated with strong polymorphonu-clear leukocyte and T lymphocyte–mediated responses to coccidioidal antigens. In most cases, a mixed inflammatory response is associated with early resolution of the infection and development of a positive delayed hypersensitivity skin test. Progressive disease is associated with weak or absent cellular immunity and skin test anergy. In most infected persons the infection is controlled after mild or inapparent illness. The disease progresses if cell-mediated immunity and consequent macrophage activation do not develop. Such immune deficits may be a result of disease (AIDS) or immuno-suppressive therapy but may occur in persons with no other known cellular immune compromise.
The central event appears to be the reaction to arthroconidia or to endospores released from ruptured spherules. Arthroconidia can be phagocytosed and killed by polymorphonu-clear leukocytes even before an adaptive immune response is mounted. The handling of endospores requires the additional participation of macrophages that do not become maxi-mally effective until activated by T lymphocyte–derived cytokines, particularly those pro-duced by the TH1 subsets. Prior to this, C. immitisendospores may be able to impair phagosome–lysosome fusion in the phagocyte.
Humoral mechanisms are not known to play any role in immunity. In fact, C. immitis is resistant to complement-mediated killing, and levels of complement-fixing antibody are in-versely related to the process of disease resolution. Persons with minimal objective indica-tions of tissue involvement (eg, lesions, radiographs) have strong T-lymphocyte responses to C. immitis antigens and little if any detectable antibody. Those with disseminated dis-ease and absent cellular immunity have high titers of antibody. Thus, the levels of antibody indicate the extent of antigenic stimulation with no known contribution to resolution of the infection.
Related Topics