Chapter: Clinical Anesthesiology: Clinical Pharmacology: Cholinesterase Inhibitors & Other Pharmacologic Antagonists to Neuromuscular Blocking Agents
Cholinesterase Inhibitors: Neostigmine
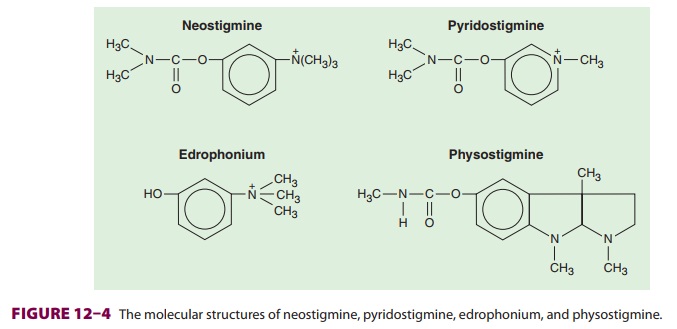
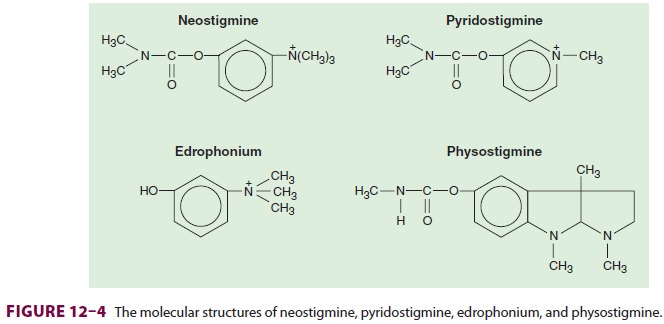
NEOSTIGMINE
Physical Structure
Neostigmine consists of a carbamate
moiety and a quaternary ammonium group (Figure 12–4). The former provides covalent
bonding to acetylcholines-terase. The latter renders the molecule lipid
insoluble, so that it cannot pass through the blood–brain barrier.
Dosage & Packaging
The maximum recommended dose of
neostigmine is 0.08 mg/kg (up to 5 mg in adults), but smaller
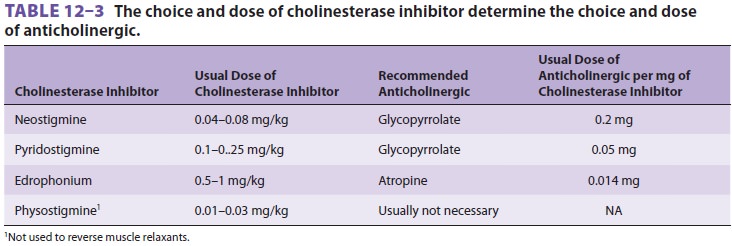
amounts often suffice and larger doses
have also been given safely ( Table 12–3). Neostigmine is most commonly
packaged as 10 mL of a 1 mg/mL solu-tion, although 0.5 mg/mL and 0.25 mg/mL
concen-trations are also available.
Clinical Considerations
The effects of neostigmine (0.04 mg/kg)
are usually apparent in 5min, peak at 10 min, and last more than 1 hr. If
reversal is not complete in 10 min after 0.08 mg/kg, the time for full recovery
of neuromus-cular function will depend on the nondepolarizing agent used and
the intensity of blockade. In practice, many clinicians use a dose of 0.04
mg/kg (or 2.5 mg) if the preexisting blockade is mild to moderate and a dose of
0.08 mg/kg (or 5 mg) if intense paralysis is being reversed. The duration of
action is prolonged in geriatric patients. Muscarinic side effects are
minimized by prior or concomitant administration of an anticholinergic agent.
The onset of action of glycopyrrolate (0.2 mg glycopyrrolate per 1 mg of
neostigmine) is similar to that of neostigmine and is associated with less
tachycardia than is experi-enced with atropine (0.4 mg of atropine per 1 mg of
neostigmine). It has been reported that neostigmine crosses the placenta, resulting
in fetal bradycardia. Thus, theoretically,
atropine may be a better choice of an anticholinergic agent than glycopyrrolate
in pregnant patients receiving neostigmine, but there is no evidence that this
makes any difference in patient outcomes. Neostigmine is also used to treat
myas-thenia gravis, urinary bladder atony, and paralytic ileus.
Related Topics