Chapter: Biotechnology Applying the Genetic Revolution: Basics of biotechnology
Chemical Structure of Nucleic Acids
CHEMICAL
STRUCTURE OF NUCLEIC ACIDS
The upcoming discussions introduce the organisms
used extensively in molecular biology and genetics research. Each of these has
genes made of DNA that can be manipulated and studied. Thus a discussion of the
basic structure of DNA is essential. The genetic information carried by DNA,
together with the mechanisms by which it is expressed, unifies every creature
on earth and is what determines our identity.
Nucleic acids include two
related molecules, deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). DNA
and RNA are polymers of subunits called nucleotides,
and the order of these nucleotides determines the information content.
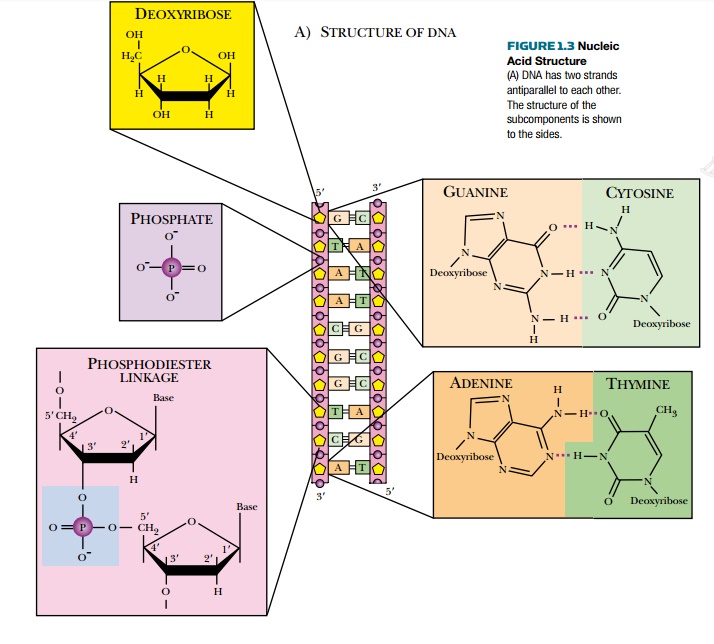
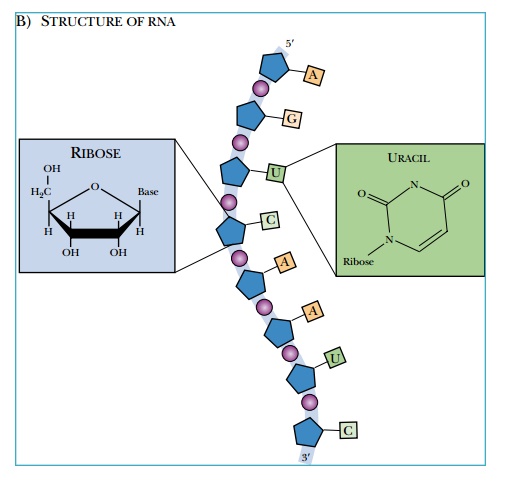
Nucleotides have three components: a phosphate
group, a five-carbon sugar, and a nitrogen-containing base (Fig. 1.3). The five-carbon
sugar or pentose is different for
DNA and RNA. DNA has deoxyribose,
whereas RNA uses ribose. These two
sugars differ by one hydroxyl group. Ribose has a hydroxyl at the 2′ position that is missing in deoxyribose. There
are five potential bases that can be attached to the sugar. In DNA, guanine,
cytosine, adenine, or thymine is attached to the sugar. In RNA, thymine is
replaced with uracil (see Fig. 1.3).
Each phosphate connects two sugars via a
phosphodiester bond. This connects the nucleotides into a chain that runs in a
5′ to 3′ direction. The 5′-OH of the sugar of one nucleotide is linked via
oxygen to the phosphate group. The 3′-OH of the sugar of the following
nucleotide is linked to the other side of the phosphate.
The nucleic acid bases jut
out from the sugar phosphate backbone and are free to form connections with
other molecules. The most stable structure occurs when another single strand of
nucleotides aligns with the first to form a double-stranded molecule, as seen
in the DNA double helix. Each base
forms hydrogen bonds to a base in the other strand. The two strands are antiparallel, that is, they run in
opposite directions with the 5′end of the first strand
opposite the 3′end of its partner and vice
versa.
The bases are of two types, purines (guanine and adenine) and pyrimidines (cytosine and thymine).
Each base pair consists of one purine connected to a pyrimidine via hydrogen
bonds. Guanine pairs only with cytosine (G-C) via three hydrogen bonds. Adenine
pairs only with thymine (A-T) in DNA or uracil (A-U) in RNA. Because an adenine-thymine
(A-T) or adenine-uracil (A-U) base pair is held together with only two hydrogen
bonds, it requires less energy to break the connection between the bases than
in a G-C pair.
The double-stranded DNA takes
the three-dimensional shape that has the lowest energy constraints. The most
stable shape is a double-stranded helix. The helix turns around a central axis
in a clockwise manner and is considered a right-handed
helix. One complete turn is 34 Å in length and has about 10 base pairs. DNA
is not static, but can alter its conformation in response to various
environmental changes. The typical conformation just described is the
B-form of DNA and is most prevalent in aqueous environments with low salt
concentrations. When DNA is in a
high-salt environment, the helix alters, making an A-form that has closer to 11 base pairs per turn. Another
conformation of DNA is the Z-form,
which has a left-handed helix with 12 base pairs per turn. In this form, the
phosphate backbone has a zigzag conformation. These two forms may be
biologically relevant under certain conditions.
DNA and RNA are both structures with
alternating phosphate and sugar residues linked to form a back-bone. Base
residues attach to the sugar and project out from the backbone. These bases can
base-pair with another strand to form double-stranded helices.
Related Topics