Transport in Plants - Cell to Cell Transport | 11th Botany : Chapter 11 : Transport in Plants
Chapter: 11th Botany : Chapter 11 : Transport in Plants
Cell to Cell Transport
Cell to Cell Transport
Cell to
cell or short distance transport covers the limited area and consists of few
cells. They are the facilitators or tributaries to the long-distance transport.
The driving force for the cell to cell transport can be passive or active
(Figure 11.1). The following chart illustrate the various types of cell to cell
transport:
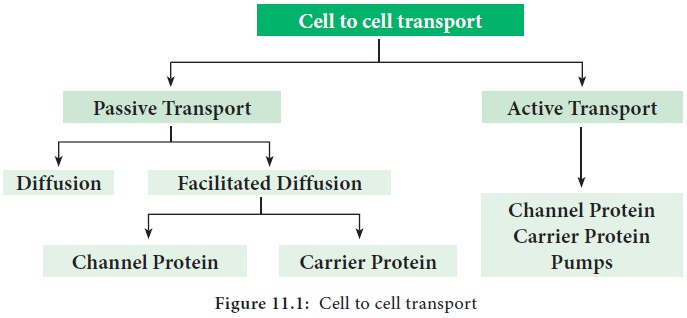
1. Passive Transport
1. Diffusion
When we
expose a lightened incense stick or mosquito coil or open a perfume bottle in a
closed room, we can smell the odour everywhere in the room. This is due to the
even distribution of perfume molecules throughout the room. This process is
called diffusion.
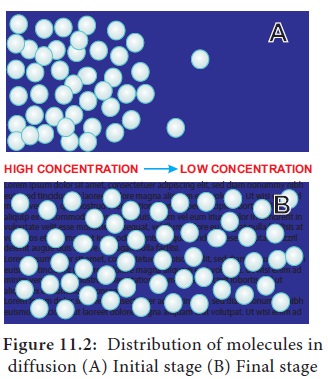
In diffusion, the movement of molecules is
continuous and random in order in all directions (Figure 11.2).
Diffusion:
The net movement of molecules from a region of their higher concentration to a
region of their lower concentration along a concentration gradient until an
equilibrium is attained.
Characteristics of diffusion
i.
It is a passive process, hence no energy
expenditure involved.
ii.
It is independent of the living system.
iii. Diffusion is obvious in gases and liquids.
iv.
Diffusion is rapid over a shorter distance but
extremely slow over a longer distance.
v.
The rate of diffusion is determined by temperature,
concentration gradient and relative density.
Significance of diffusion in Plants
i. Gaseous exchange of O2 and CO2
between the atmosphere and stomata of leaves takes place by the process of
diffusion. O2 is absorbed during respiration and CO2 is
absorbed during photosynthesis.
ii.
In transpiration, water vapour from intercellular
spaces diffuses into atmosphere through stomata by the process of diffusion.
iii.
The transport of ions in mineral salts during
passive absorption also takes place by this process.
2. Facilitated Diffusion
Cell
membranes allow water and nonpolar molecules to permeate by simple diffusion.
For transporting polar molecules such as ions, sugars, amino acids, nucleotides
and many cell metabolites is not merely based on concentration gradient. It
depends on,
i Size of molecule: Smaller
molecules diffuse faster.
![]()
ii. Solubility of the molecule: Lipid soluble
substances easily and rapidly pass through the membrane. But water soluble
substances are difficult to pass through the membrane. They must be facilitated
to pass the membrane.
Types of Membrane Permeability
A solution is made up of solute particles dissolved in a solvent and the permeability of the above components depends on the nature of cell membranes, which is given below:
Impermeable: Inhibit the movement of both solvent and solute molecules. Example: Suberised, cutinesed or liginifid cell walls.
Permeable: They allow diffusion of
both solvent and solute molecules through them. Example: Cellulosic cell wall.
Semi permeable: Semi permeable allow diffusion of solvent molecules but do not allow the passage of solute molecule. Example: Parchment paper.
Selectively permeable: All bio membranes
allow some solutes to pass in addition to the solvent molecules. Example:
Plasmalemma, tonoplast, and membranes of cell organelles.
In
facilitated diffusion, molecules cross the cell membrane with the help of
special membrane proteins called transport proteins, without the expenditure of
ATP.
There are
two types of transport proteins present in the cell membrane. They are channel
protein and a carrier protein.
I. Channel Protein
Channel
protein forms a channel or tunnel in the cell membrane for the easy passage of
molecules to enter the cell. The channels are either open or remain closed.
They may open up for specific molecules. Some channel proteins create larger pores
in the outer membrane. Examples: Porin and Aquaporin.
i.
Porin
Porin is
a large transporter protein found in the outer membrane of plastids,
mitochondria and bacteria which facilitates smaller molecules to pass through
the membrane.
ii.
Aquaporin
Aquaporin
is a water channel protein embedded in the plasma membrane. It regulates the
massive amount of water transport across the membrane (Figure 11.3). Plants
contain a variety of
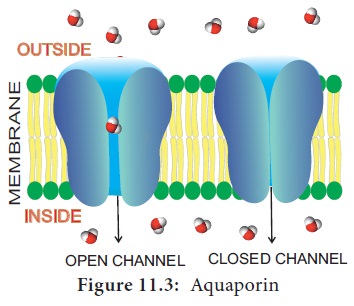
Over 30 types of aquaporins are known from maize. Currently, they are also recognized to transport substrates like glycerol, urea, CO2, NH3, metalloids, and Reactive Oxygen Species (ROS) in addition to water. They increase the permeability of the membrane to water. They confer drought and salt stress tolerance.
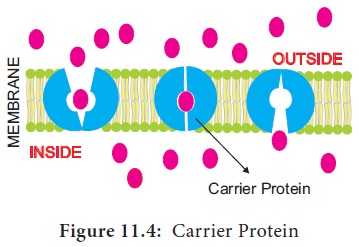
II. Carrier Protein
Carrier
protein acts as a vehicle to carry molecules from outside of the membrane to
inside the cell and vice versa (Figure 11.4). Due to association with molecules
to be transported, the structure of carrier protein gets modified until the
dissociation of the molecules.
There are 3 types of carrier proteins classified on the basis of handling of molecules and direction of transport (Figure 11.5). They are,
i) Uniport
ii) Symport
iii) Antiport.
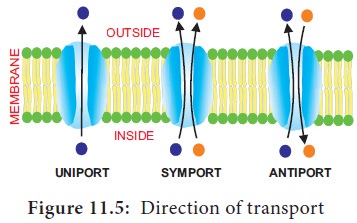
i.
Uniport: In this molecule of a single type
move across a membrane independent of other molecules in one direction.
ii.
Symport or co-transport: The
term
symport is used to denote an integral membrane protein that
simultaneously transports two types of molecules across the membrane in the
same direction.
iii. Antiport
or Counter Transport: An antiport is an integral membrane transport protein that simultaneously
transports two different molecules, in opposite directions, across the
membrane.
2. Active Transport
The main
disadvantage of passive transport processes like diffusion is the lack of
control over the transport of selective molecules. There is a possibility of
harmful substances entering the cell by a concentration gradient in the
diffusion process. But selective permeability of cell membrane has a great
control over entry and exit of molecules. Active transport is the entry of
molecules against a concentration gradient and an uphill process and it needs
energy which comes from ATP. Passive transport uses kinetic energy of molecules
moving down a gradient whereas, active transport uses cellular energy to move
them against a gradient. The transport proteins discussed in facilitated
diffusion can also transport ions or molecules against a concentration gradient
with the expenditure of cellular energy as an active process. Pumps use a
source of free energy such as ATP or light to drive the thermodynamically
uphill transport of ions or molecules. The pump action is an example of active
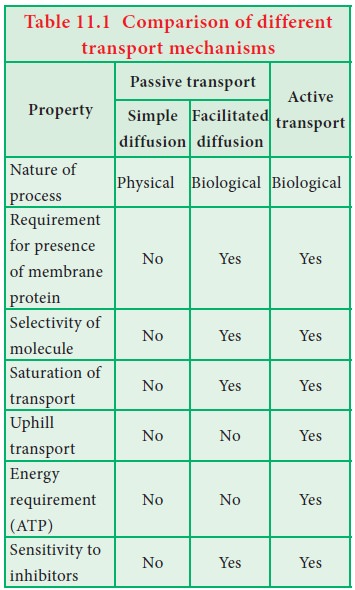
transport. Example: Na+-K+-ATPase pump (Table 11.1).
Check your grasp!
What are the similarities and differences between co- transport and counter transport?
Solution:
Similarity: In both system two molecules are involved for the
unidirectional transport.
Difference: In co-transport, two molecules are transported
together whereas, in counter transport two molecules are transported in
opposite direction to each other.
Related Topics