Chapter: Microbiology and Immunology: Bacteriology: Neisseria
Cell Wall Components and Antigenic Properties - Neisseria gonorrhoeae
Cell Wall Components and Antigenic Properties
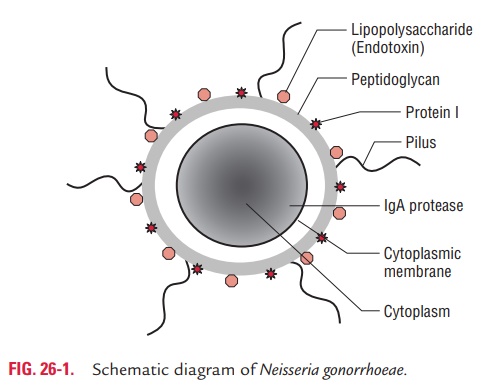
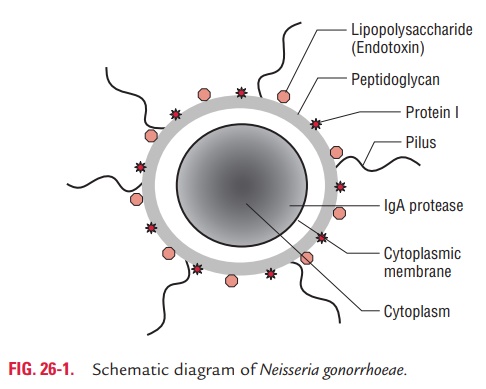
The cell wall of N. gonorrhoeae like any other Gram-negative bacteria consists of three layers: outer cell surface, middle pep-tidoglycan layer, and inner cytoplasmic membrane. These con-tain following proteins (Fig. 26-1).
◗ Outer membrane proteins
The outer membrane proteins (OMP) are present in the outer membrane. They mediate the uptake of iron essential for growth and metabolism of the cocci. They promote intake of iron by binding hemoglobin, transferrin, and lactoferrin. These proteins are of three types:
· The Por proteins
· The Opa proteins
· The Rmp proteins
The Por proteins: The Por proteins, earlier known as proteinI, are porin proteins that form pores or channels in the outer membranes. Por proteins are of two types: Por-A and Por-B, each with a variety of antigenic variations. Strains producing Por-A proteins are commonly associated with disseminated dis-ease because these proteins prevent killing of gonococci in the serum by the serum complement components. The antigenic variations observed in Por proteins form the basis for the sero-type classification of N. gonorrhoeae.
The Opa proteins: These proteins, also known as opacityprotein, were formerly known as protein II. These proteins are found in the membrane and mediate adherence of the bacteria to each other, and also to the eukaryotic cells. Strains produc-ing Opa proteins produce opaque colonies in culture.
The Rmp proteins: These proteins, also known as reductionmodifiable proteins, were formerly known as protein III. These are proteins found in the outer membrane of gonococci and lead to the production of antibodies that block serum bacteri-cidal activity against gonococci.
◗ Other important gonococcal proteins
Lipo-oligosaccharide (LOS) is another major antigen present in the cell wall of the bacteria. This antigen consists of lipid A and oligosaccharide similar to that of lipopolysaccharide (LPS) of Gram-negative bacteria. However, LOS does not show antigenic variation as found in LPS. LOS possesses endotoxic activity. IgA1 protease and beta-lactamase are the other impor-tant proteins. IgA1 protease degrades secretory IgA1, whereas beta-lactamase degrades beta-lactam rings in the penicillin.
Related Topics