Chapter: Mechanical : Advanced IC Engines : Engine Exhaust Emission Control
Catalytic convertor
Catalytic convertor:
The
catalytic converters used in spark-ignition engines consist of an active
catalytic material in a specially designed metal casing which directs the
exhaust gas flow through the catalyst bed. The active material employed for CO
and HC oxidation or NO reduction (normally noble metals, though base metals
oxidescan be used) must be distributed over a large surface area so that the
mass transfer characteristics between the gas phase and the active catalyst
surface are sufficient to allow close to 100 percent conversion with high
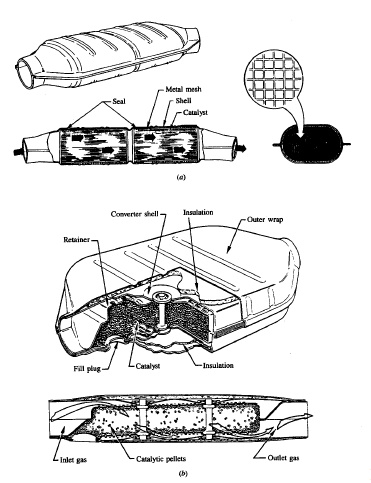
catalytic activity. The two configurations commonly used are shown in Fig.
11-53. One systememploys a ceramic honeycomb structure or monolith held in a
metal can in theexhaust stream. The active (noble metal) catalyst material is
impregnated into ahighly porous alumina washcoat about 20 pm thick that is
applied to the passagewaywalls. The typical monolith has square-cross-section
passageways with inside dimensions of @1 mm
separated by thin (0.15 to 0.3 mm)
porous walls.
The
number of passageways per square centimetre varies between about 30 and60. The
washcoat, 5 to 15 percent of the weight of the monolith, has a surfacearea of
100 to 200 m2/g. The other converter design uses a bed of sphericalceramic
pellets to provide a large surface area in contact with the flow. With
pellet
catalysts, the noble metal catalyst is impregnated into the highly
poroussurface of the spherical alumina pellets (typically 3 mm diameter) to a
depth ofabout 250 pm. The pellet material is chosen to have good crush and
abrasion resistance after exposure to temperatures of order 1000•C. The gas
flow is directed down through the bed as shown to provide a large flow area and
low pressure drop. The gas flow is turbu lent which results in high
mass-transfer rates; in the monolith catalyst passageways, it is laminar.
Related Topics