Chapter: Clinical Anesthesiology: Anesthetic Management: Renal Physiology & Anesthesia
Anesthesia: The Nephron
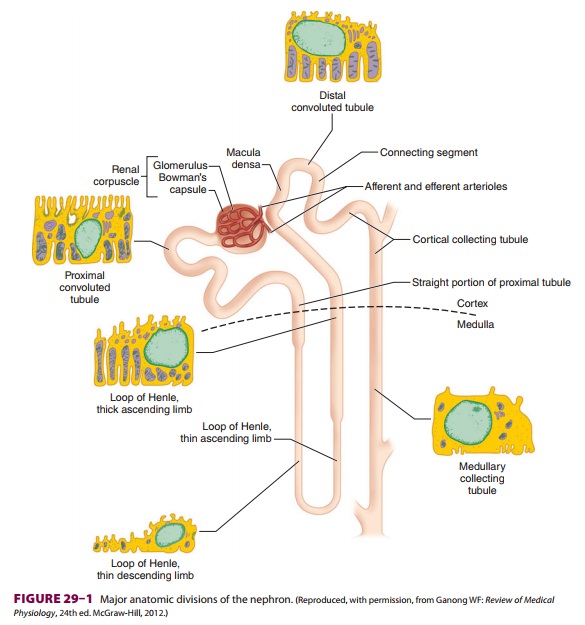
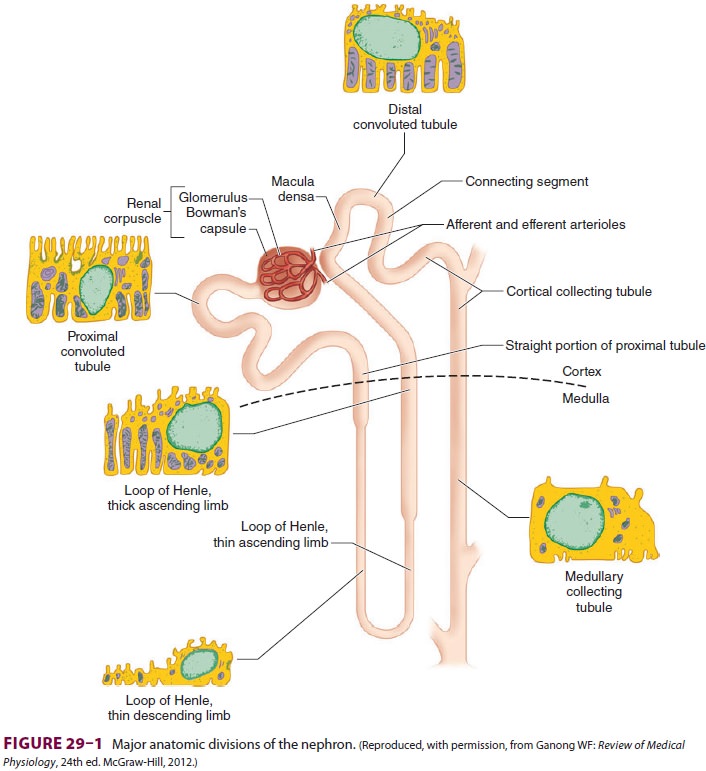
The Nephron
Each kidney is made up of approximately 1 million functional
units called nephrons. Anatomically, a nephron consists of a tortuous tubule
with at least six specialized segments. At its proximal end (the renal corpuscle, composed of a
glomerulus anda Bowman’s capsule), an ultrafiltrate of blood is formed, and as
this fluid passes through the neph-ron, its volume and composition are modified
by both the reabsorption and the secretion of solutes. The final product is
eliminated as urine.
Nephrons are classified as cortical
or juxta-medullary , and the renal
corpuscles ofall nephrons are located in the renal cortex. The six major
anatomical and functional divisions of the nephron are the renal corpuscle, the
proximal con-voluted tubule, the loop of Henle, the distal renal tubule, the
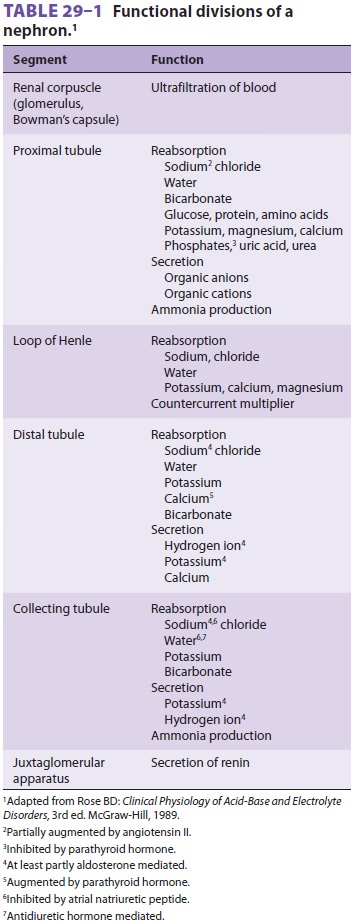
collecting tubule, and the juxtaglomeru-lar apparatus (Figure29–1
and Table29–1).
The Renal Corpuscle
Each renal corpuscle contains a
glomerulus, which is composed of tuf ts of capillaries that jut into Bowman’s
capsule, providing a large surface area for the filtration of blood. Blood flow
is provided by a single afferent arteriole and is drained by a sin-gle efferent
arteriole . Endothelial cells of the glomeruli are separated from the
epithelial cells of Bowman’s capsule only by their fused base-ment membranes.
The endothelial cells are perfo-rated with relatively large fenestrae (70–100
nm), but the epithelial cells interdigitate tightly with one another, leaving
relatively small filtration slits (about 25 nm). The two cell types with their
base-ment membranes provide an effective filtration bar-rier to cells and
large-molecular-weight substances. This barrier has multiple anionic sites that
give it a net negative charge, favoring filtration of cations relative to
anions. A third cell type, called intraglo-merular mesangial cells, is located
between the base-ment membrane and epithelial cells near adjacent capillaries.
These contractile cells regulate glomeru-lar blood flow and also exhibit
phagocytic activity. They secrete various substances, absorb immune complexes,
and contain contractile proteins that respond to vasoactive substance.
Mesangial cells contract, reducing glomerular filtration, in response to
angiotensin II, vasopressin, norepinephrine, his-tamine, endothelins,
thromboxane A2, leukotrienes (C4 and D4), prostaglandin F2, and
platelet-acti-vating factor. They relax, thereby increasing glo-merular
filtration, in response to atrial natriuretic peptide (ANP), prostaglandin E2,
and dopaminer-gic agonists.
Glomerular filtration pressure (about 60 mm Hg) is normally
approximately 60% of mean arte-rial pressure and is opposed by both plasma
oncotic pressure (about 25 mm Hg) and renal interstitial pressure (about 10 mm
Hg). Afferent and efferent arteriolar tone are both important in determin-ing
glomerular filtration pressure: filtration pres-sure is directly proportional
to efferent arteriolar tone but inversely proportional to afferent tone.
Approximately 20% of plasma is normally filtered as blood passes through the
glomerulus.
The Proximal Tubule
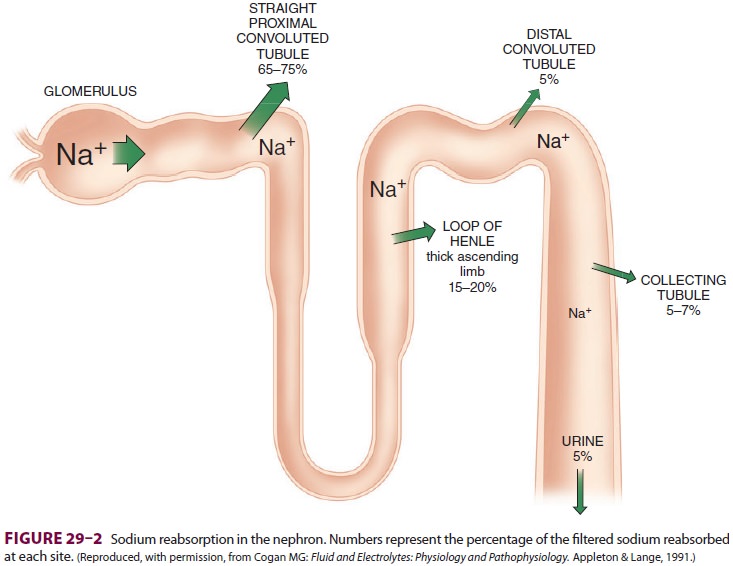
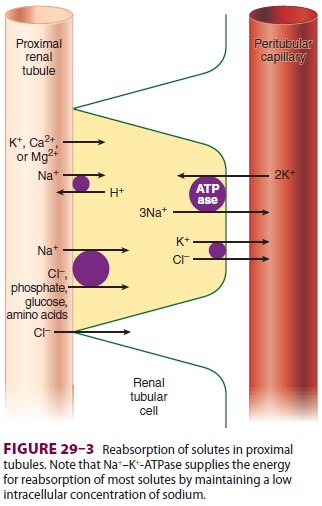
Of the ultrafiltrate formed in Bowman’s capsule 65–75% is normally reabsorbed isotonically (pro-portional amounts of water and sodium) in the proximal renal tubules (Figure29–2). To be reab-sorbed, most substances must first traverse the tubular (apical) side of the cell membrane, and then cross the basolateral cell membrane into the renal interstitium before entering peritubular capillaries. The major function of the proximal tubule is Na + reabsorption. Sodium is actively transported out of proximal tubular cells at their capillary side by membrane-bound Na+–K+-adenosine triphospha-tase (Na+–K+-ATPase) (Figure29–3). The result-ing low intracellular concentration of Na + allows passive movement of Na+ down its gradient from tubular fluid into epithelial cells. Angiotensin II and norepinephrine enhance Na+ reabsorption in the
early proximal tubule. In contrast, dopamine and fenoldopam
decrease the proximal reabsorption of sodium via D 1-receptor activation.
Sodium reabsorption is coupled with the reabsorption of other solutes and the secretion of H+ (Figure 29–3). Specific carrier proteins use the low concentration of Na+ inside cells to transport phosphate, glucose, and amino acids. The net loss of intracellular positive charges, the result of Na +–K+-ATPase activity (exchanging 3Na + for 2K+), favors the absorption of other cations (K +, Ca2+, and Mg2+). Thus, the Na+–K+-ATPase at the basolateral side of the renal cells provides the energy for the reab-sorption of most solutes. Sodium reabsorption at the luminal membrane is also coupled with coun-tertransport (secretion) of H +. The latter mecha-nism is responsible for reabsorption of 90% of the filtered bicarbonate ions (see Figure 50–3). Unlike other solutes, chloride can traverse the tight junc-tions between adjacent tubular epithelial cells, and accordingly, is passively resorbed via its concentra-tion gradient. Active chloride reabsorption may also take place as a result of a K+–Cl– cotransporter that extrudes both ions at the capillary side of the cell membrane (Figure 29–3). Water moves pas-sively out the proximal tubule along osmotic gra-dients. Apical membranes of epithelial cells contain specialized water channels, composed of a mem-brane protein called aquaporin-1, that facilitate water movement.
The proximal tubules are capable of secreting organic cations
and anions. Organic cations such as creatinine, cimetidine, and quinidine may
share the same pump mechanism and thus can compete for excretion with one
another. Organic anions such as urate, ketoacids, penicillins, cephalosporins,
diuret-ics, salicylates, and most radiocontrast dyes also share common
secretory mechanisms. Both pumps probably play a major role in the elimination
of many circulating toxins. Low-molecular-weight proteins, which are filtered
by glomeruli, are normally reab-sorbed by proximal tubular cells, to be
metabolized intracellularly.
The Loop of Henle
The loop of Henle consists of descending and ascending portions.
They are responsible for
maintaining a hypertonic medullary interstitium and also
indirectly provide the collecting tubules with the ability to concentrate
urine. The thin descending segment is a continuation of the proxi-mal tubule
and descends from the renal cortex into the renal medulla. In the medulla, the
descending portion acutely turns back upon itself and rises back up toward the
cortex as the ascending por-tion. The ascending portion consists of a
function-ally distinct, thin ascending limb, a medullary thick ascending limb,
and a cortical thick ascending limb (Figure 29–1). Cortical nephrons have relatively short loops of Henle which extend
only into the more superficial regions of the renal medulla and often lack a
thin ascending limb. Juxtamedullary
nephrons, which have renal corpuscles located near the renal medulla, possess
loops of Henle that project deeply into the renal medulla. Cortical nephrons
outnumber juxtamedullary nephrons by approximately 7:1.Only 25–35% of the ultrafiltrate formed in Bowman’s
capsule normally reaches the loop of Henle. Once there, 15–20% of the filtered
sodium load is normally reabsorbed in the loop of Henle. With the notable
exception of the ascending thick segments, solute and water reabsorption in the
loop of Henle is passive and follows concentration and osmotic gradients,
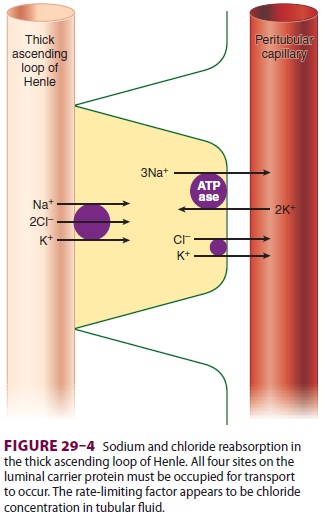
respectively. In the ascending thick segment, however, Na+ and Cl − are
reabsorbed in excess of water; moreover, Na+ reabsorption in this part of the
nephron is directly coupled to both K+ and Cl − reabsorption (Figure 29–4), and
[Cl−] in tubular fluid appears to be the rate-limiting factor. Active Na+
reabsorption still results from
Na+–K+-ATPase
activity on the capillary side of epi-thelial cells.
Unlike the descending limb and the thin
ascending limb, the thick parts of the ascending limb are impermeable to water.
As a result, tubular fluid flowing out of the loop of Henle is hypotonic (100–
200 mOsm/L) and the interstitium surrounding the loop of Henle is therefore
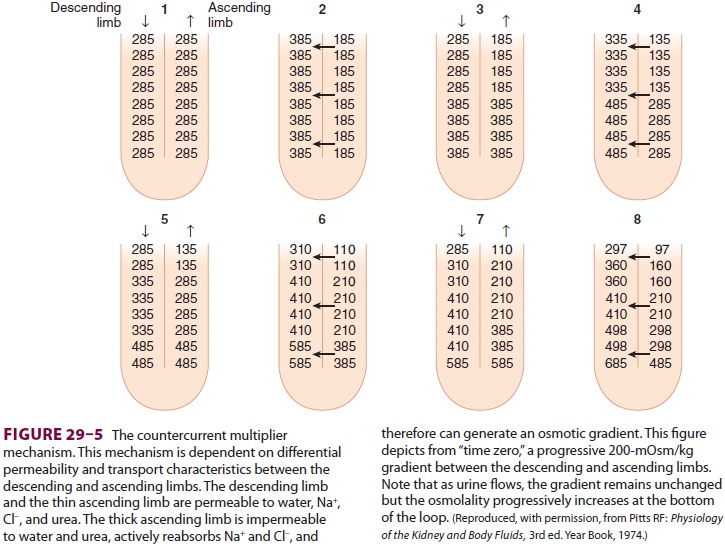
hypertonic. A countercur-rent multiplier mechanism is established such that
both the tubular fluid and medullary interstitium become increasingly
hypertonic with increasing depth into the medulla ( Figure 29–5). Urea
con-centrations also increase within the medulla and contribute to the
hypertonicity. The countercurrent mechanism includes the loop of Henle, the
cortical and medullary collecting tubules, and their respec-tive capillaries
(vasarecta).
The thick ascending loop of Henle is also an important site for
calcium and magnesium reabsorption, and parathyroid hormone may aug-ment
calcium reabsorption at this location.
The Distal Tubule
The distal tubule receives hypotonic fluid from the loop of Henle and is normally responsible for only minor modifications of tubular fluid. In contrast to more proximal portions, the distal nephron has very tight junctions between tubular cells and is relatively impermeable to water and sodium. It can there-fore maintain the gradients generated by the loop of Henle. Sodium reabsorption in the distal tubule normally accounts for only about 5% of the filtered sodium load. As in other parts of the nephron, the energy is derived from Na+–K+-ATPase activity on the capillary side, but on the luminal side Na + is reabsorbed by an Na +–Cl− carrier. Sodium reabsorp-tion in this segment is directly proportional to Na+ delivery. The distal tubule is the major site of para-thyroid hormone–and vitamin D–mediated calcium reabsorption.
The
latter portion of the distal tubule is referred to as the connecting segment. Although it is also involved in
hormone-mediated calcium reabsorp-tion, unlike more proximal portions, it
participates in aldosterone-mediated Na+
reabsorption.
The Collecting Tubule
The collecting tubule can be divided into cortical and medullary
portions. Together, they normally account for the reabsorption of 5–7% of the
filtered sodium load.
A. Cortical Collecting Tubule
Th is part of the
nephron consists of two cell types:
principal
cells (P cells), which primarily secrete potassium and participate in
aldosterone-stimulated Na+
reabsorption, and (2) intercalated cells (I cells), which are responsible for
acid–base regulation. Because P cells reabsorb Na+
via an electrogenic pump, either Cl−
must also be reabsorbed or K+
must be secreted to maintain electroneutrality. Increased intracellular [K +]
favors K+ secretion. Aldosterone
enhances Na +–K+-ATPase
activity in this part of the nephron by increasing the number of open K+
and Na+ channels in the
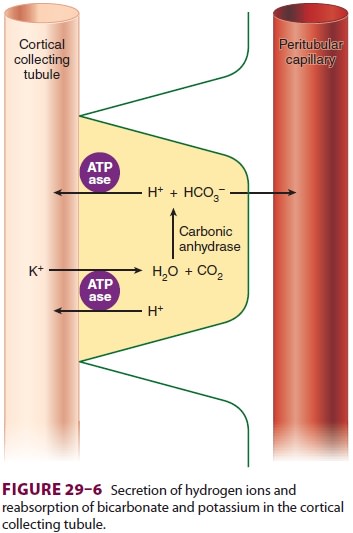
luminal membrane. Aldosterone also enhances the H +-secreting
ATPase on the lumi-nal border of I cells (Figure29–6). I cells
addition-ally have a luminal K +–H+-ATPase
pump, which reabsorbs K+
and secretes H+, and some I cells are
capable of secreting bicarbonate ion in response to large alkaline loads.
B. Medullary Collecting Tubule
The medullary collecting tubule courses down from the cortex through the hypertonic medulla before joining collecting tubules from other nephrons to form a single ureter in each kidney. This part of the collecting tubule is the principal site of action for antidiuretic hormone (ADH), also called arginine vasopressin (AVP). ADH stimulates the expression of a water channel protein, aquaporin-2, in the cell membrane. The permeability of the luminal mem-brane to water is entirely dependent on the presence of ADH . Dehydration increases ADH secretion, rendering the luminal membrane permeable to water. As a result, water is osmotically drawn out of the tubular fluid passing through the medulla, resulting in production of concentrated urine (up to 1400 mOsm/L). Conversely, adequate hydration suppresses ADH secretion, allowing fluid in the collecting tubules to pass through the medulla relatively unchanged and to remain hypo-tonic (100–200 mOsm/L). This part of the nephron is responsible for acidifying urine; the hydrogen ions
The Juxtaglomerular Apparatus
This small organ within each nephron
consists of a specialized segment of the afferent arteriole, con-taining
juxtaglomerular cells within its wall, and the end of the thick, ascending
cortical segment of the loop of Henle, the macula densa ( Figure29–7).
Juxtaglomerular cells contain the enzyme renin and are innervated by the
sympathetic nervous system. Release of renin depends on β1-adrenergic sympa-thetic stimulation,
changes in afferent arteriolar wall
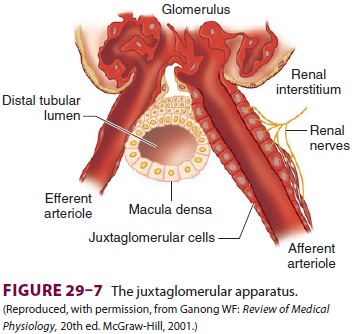
pressure , and changes in chloride flow past the macula densa.
Renin released into the bloodstream catalyzes the conversion of angio-tensinogen,
a protein synthesized by the liver, to angiotensin I. This inert decapeptide is
then rapidly converted, primarily in the lungs, by angiotensin-converting
enzyme (ACE) to form the octapeptide angiotensin II. Angiotensin II plays a
major role in blood pressure regulation
and aldosterone secretion . Proximal renal tubular cells have converting
enzyme as well as angiotensin II receptors. Moreover, intrarenal formation of
angiotensin II enhances sodium reab-sorption in proximal tubules. Some
extrarenal pro-duction of renin and angiotensin II also takes place in the
vascular endothelium, the adrenal glands, and the brain.
Related Topics